 |
 |


- Search
| Int Neurourol J > Volume 28(Suppl 1); 2024 > Article |
|
ABSTRACT
Purpose
The prevalence of lower urinary tract symptoms (LUTS), characterized by problems regarding storage and/or voiding of urine, is known to significantly increase with age. Effective communication between the lower urinary tract and the central nervous system (CNS) is essential for the optimal function of this system, and heavily relies on the efficient interaction between the bladder urothelium and the afferent nerve fibers situated in close proximity to the urothelium within the lamina propria.
Methods
We aimed to quantify aging-related differences in the expression of calcitonin gene-related peptide (CGRP, an established marker for sensory nerve fibers) in the trigonal mucosal layers of young (3–4 months) and aged (25–30 months) rats. We evaluated trigonal tissue from 3 animals per age group. Tissue was serially sectioned at 10 μm and stained for CGRP. Images were taken along the full length of the tissue. For each image we computed the total CGRP-positive area (μm2) and the median value for each animal was used for further analysis.
Results
Upon statistical analysis the aged rats show a significantly lower CGRP-positive area compared to young rats (P=0.0049). These results indicate that aging has a negative effect on the area of CGRP-positive signal in the trigone.
Conclusions
The structural and functional integrity of the sensory web in the trigonum of rats is negatively affected by the aging process, potentially leading to impaired communication between the bladder urothelium the CNS. Consequently, these perturbations in the sensory system may contribute to the pathogenesis or exacerbation LUTS.
- In this study, we quantified aging-related deterioration of afferent nerve fibers in the trigonal mucosa of rats.
- We argue that these changes might significantly impact lower urinary tract sensation and control and potentially play an important role in the increased prevalence of lower urinary tract symptoms in older adults.
With advancing age, there is a noticeable rise in the prevalence of lower urinary tract symptoms (LUTS), which are characterized by challenges related to storage and/or voiding of urine [1]. These symptoms encompass issues such as frequent urination, urgency, difficulty initiating urination, nocturia, and incomplete emptying of the bladder [2, 3]. The impact of LUTS on an individual’s quality of life and overall well-being underscores the importance of understanding and addressing this age-associated phenomenon. The aging process can affect multiple components of the lower urinary tract, leading to various dysfunctions. Lower urinary tract components that may become dysfunctional with aging include the urothelial lining, which may become more susceptible to irritation and inflammation; the musculature, which can lose its strength and tone, resulting in decreased bladder contractility; the stromal tissue, which may undergo structural changes affecting bladder integrity; and the neural pathways, which can become less efficient at coordinating bladder function. These aging-related alterations in lower urinary tract physiology can contribute to an increased risk of LUTS [4-6].
The maintenance of a homeostatic equilibrium relies heavily on the optimal execution of viscerosceptive processes [7]. These processes encompass the body’s ability to detect and respond to signals originating from organs including the cardiac, gastrointestinal and urinary systems. There is a continuous exchange of information between the peripheral (PNS) and central nervous system (CNS) to ensure that internal bodily cues are accurately perceived and appropriately acted upon [8]. This intricate interplay is essential for regulating various bodily functions, such as the control of blood pressure, digestion, and urinary bladder function. Optimal coordination of these processes facilitates homeostasis by ensuring that the body appropriately adapts to changing demands and maintains a state of equilibrium.
Interestingly, the process of aging is associated with a decrease in visceroceptive awareness [9, 10]. This means that as we age, we may become less attuned to the signals our body sends us regarding various bodily functions, such as hunger, fullness, or the need to urinate. This decline in awareness can have significant implications for our overall health and well-being.
Currently, our understanding of the specific mechanisms by which the aging process detrimentally affects lower urinary tract functioning is largely incomplete. A more thorough understanding of these mechanisms could provide valuable information about aging-related changes in lower urinary tract function, and will be essential for the further development of interventions that enhance the well-being of the aging population.
The correct execution of communication between the lower urinary tract and the CNS is crucial for optimal bladder functioning. This communication heavily relies upon the seamless interaction between the bladder urothelium, the innermost layer of the bladder wall, and afferent nerve fibers situated within the lamina propria, a connective tissue layer closely adjacent to the urothelium [11]. While bladder afferent sensory nerve fibers are distributed throughout the bladder, the bladder trigone serves as a sensory “hotspot” and contains a higher density of afferent nerve fibers compared to other bladder regions [12]. It is likely that dysfunction in this system is associated with the reported aging-related increase in the prevalence of LUTS.
In the current study we quantified aging-related differences in the expression of calcitonin gene-related peptide (CGRP, an established marker for sensory nerve fibers) in the trigonal mucosal layers of young (3–4 months) and aged (25–30 months) rats. We hypothesized that trigonal mucosa obtained from aged rats shows significantly less CGRP expression compared to young rats.
This study utilized female young, though mature (3–4 months old) and aged (25–30 months old) Fischer 344 rats ([F344] Charles River, Wilmington, MA, USA; National Institute on Aging rodent colony, Charles River Laboratories, Wilmington, MA, USA). Animals were kept in standard housing with 12-hour light/dark cycles and ad libitum access to food and water. Bladder tissue (N=3 per age group) was collected after sacrificing animals by exsanguination during isoflurane anesthesia. All animal procedures received ethical approval from the University of Pittsburgh Institutional Animal Care and Use Committee.
The bladder was cut open longitudinally and pinned flat with the lumen side upwards. Tissue was post-fixed for 60 minutes in 4% paraformaldehyde (PFA) in phosphate-buffered saline (PBS), immersed in 30% sucrose for >24 hours. The trigonal region between the ureteral orifices and the urethra was isolated, embedded in optimum cutting temperature compound medium (Tissue-Tek OCT, Sakura Finetek, Torrance, CA, USA) and stored at -80°C until cutting. Tissue was then serially sectioned at 10-μm thickness and mounted on glass slides (Fisher Scientific, Pittsburgh, PA, USA) with 3 sections per slide. Sections were air dried >1 hour, washed with PBS, incubated with permeabilizing block solution (5% normal donkey serum, 1% bovine serum [Sigma-Aldrich, St. Louis, MO, USA] and 0.2% Triton X-100) for 1 hour, followed by incubation with primary antibody (anti-CGRP, 1:2,000, Sigma-Aldrich) overnight at 4°C. We used validated antibodies that detect CGRP and target CGRP in human, rat, and mouse samples. Negative controls were included for each animal in which exposure of the tissue to the primary antibody was omitted. The following day, slides were washed with PBS and incubated with secondary Cy3 goat anti-rabbit (Alexa Fluor 555, 1:500) in blocking solution for 2 hours at room temperature. DAPI (1:2,500) 15 minutes at room temperature was then used to counterstain the nuclei. Slides were post fixated with 4% PFA for 10 minutes and washed with PBS. Slides were then cover slipped using mounting medium (Immu-Mount, Fisher Scientific) and dried >24 hours at room temperature in the dark. Control experiments included omission of primary antibody from blocking solution.
Stacked images were taken so that the full thickness (10 μm) and complete length of the lamina propria of each section was fully imaged. The stacked images were subsequently saved as 2-dimensional images containing data from the full 10-μm thickness of each section. Using an automated approach, images (N=160) were binarized in ImageJ using the triangle algorithm for optimal thresholding and median filtering was applied to reduce single pixel noise. A region of interest detection threshold was applied to detect a minimum of 10 connected CGRPpositive pixels. Each CGRP-positive area of 10 or more connected pixels was included, and for each image we computed the total CGRP-positive area (μm2). The median value for each animal was subsequently used for further analysis.
Presence and distribution of CGRP was evaluated in trigonal tissue obtained from young and aged rats in order to evaluate aging-associated changes in afferent nerve fibers in the bladder trigone.
Negative controls consistently exhibited no detectable fluorescence signal when subjected to the appropriate excitation and emission wavelengths. Representative images of CGRP expression in young and aged trigonal mucosa are shown in Fig. 1A and B. Upon visual inspection it can be observed that afferent nerve fiber endings in the trigone of aged rats appear to penetrate less deeply into the urothelium (white arrows in the figure indicate fine nerve fiber endings penetrating the urothelium in the trigone of young animals). Furthermore, fibers appear more fragmented across the full 10-μm thickness of the sections.
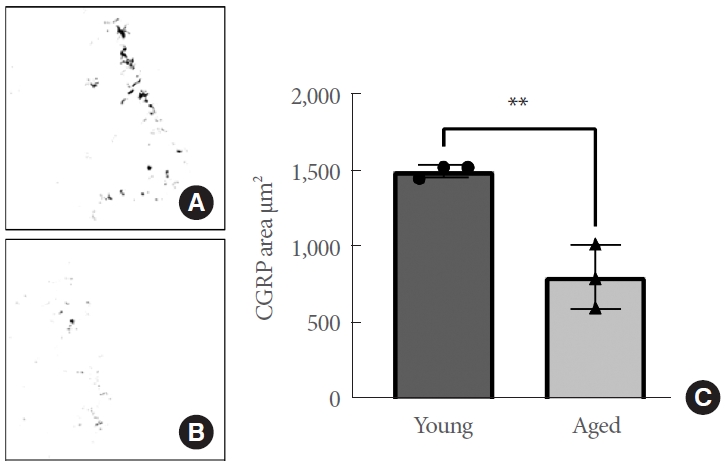
The median area of CGRP-positive pixels for all images (N= 160) was computed and was observed to be highly similar across animals of the same age group and showed a strong normal distribution. Furthermore, variances did not differ between age groups. Upon statistical analysis, the trigonal mucosa of aged animals shows a significantly lower CGRP-positive area compared to young animals (N=3 per age group, P=0.0049) (Fig. 2). These results indicate that aging has a negative effect on the area (μm2) of CGRP-positive signal in the bladder trigone. The structural and functional integrity of the web of sensory nerves in the trigonum of aged animals is significantly affected, and communication between the bladder urothelium and the CNS is likely compromised as a result.
In the current study, we evaluated the effects of aging on trigonal sensory innervation. We assessed the presence and distribution of CGRP in trigonal tissue obtained from young and aged rats. Our findings revealed that aging negatively affects the presence of fine afferent nerve fiber endings in the urothelium upon visual inspection. Additionally, quantitative assessment of CGRP-positive areas (μm2) in the bladder trigone of young and aged rats revealed a significant aging-associated decrease in CGRP-positive area. These findings suggest that sensory signaling capabilities from the urothelium are likely compromised in aging. This observation may negatively impact the communication between the lower urinary tract and the CNS, and could be associated with aging-related decreases in visceroceptive awareness.
Under normal physiological circumstances, bladder afferent nerve fibers are mainly observed in the lamina propria, just underneath the urothelium, with numerous fine nerve fiber endings extending into the urothelial layers [13, 14]. Our observation that aging negatively affects fine afferent nerve fiber endings in the trigonal urothelium, as well as the overall presence of afferent nerve fibers in the lamina propria, illustrates the disruptive impact of the aging process on trigonal sensory innervation. These delicate nerve fibers play an essential role in transmitting sensory information from the bladder to CNS, ensuring optimal regulation of normal bladder function, and the reported negative changes in afferent nerve fibers likely have a profound impact on bladder functioning. Pathophysiological situations including urgency, increased voiding frequency, nocturia, urinary incontinence, and bladder pain syndrome have been associated with maladaptive changes in bladder afferent signaling [15-17]. These maladaptive changes may encompass alterations in nerve sensitivity, aberrant reflexes, and abnormal neurotransmitter release, contributing to the diverse spectrum of LUTS observed in these conditions. Aging has been associated with an increased prevalence of a multitude of LUTS [2, 3], and the reported results suggest that the deterioration of optimal bladder afferent signaling, resulting from negative changes in trigonal afferent nerve fibers, is a likely contributor to agingrelated lower urinary tract dysfunction. Moreover, our quantitative assessment of CGRP-positive areas in the bladder trigone of both young and aged rats provides a tangible measure of the age-associated decline in sensory nerve fiber density, reinforcing the concept that aging detrimentally affects the structural integrity of trigonal sensory innervation.
Aging-associated effects on peripheral nerve functioning are relatively well established and research indicates that aging has a substantial impact on the PNS, resulting in the loss of nerve fibers, structural abnormalities, and functional decline [18]. Aging is associated with alterations in the expression of neurotrophic factors such as nerve growth factor (NGF), and the balance between the expression of NGF and its precursor proNGF [19, 20]. Neurotrophic factors including NGF and brain-derived nerve factor have been proposed to fulfill regulatory roles in the execution of optimal bladder functioning [21, 22]. Neurotrophic factors are essential for supporting the growth, maintenance, and survival of nerve fibers. A potential dysregulation of neurotrophic factors in the urothelium and lamina propria may underlie the reported deterioration of afferent nerve fibers. Furthermore, the aging process leads to increases in oxidative stress in the bladder wall, which may negatively impact the signaling capacities of the urothelium [6, 23]. Prolonged exposure to oxidative stress may lead to degeneration of nerve fibers in the lamina propria and may stimulate inflammatory responses which can further contribute to nerve damage and sensory nerve fiber dysfunction. A study investigating the effects of aging on epidermal innervation and intra-epidermal nerve fiber density evaluated human skin biopsies obtained from different regions including the leg, thigh, and trunk [24]. Significant aging-related differences could only be established for the trunk region, suggesting that there are regional variations in the impact of aging on nerve fibers. The observation that nerve fibers in the abdominal region are negatively affected by the aging process are in line with the results reported in the current study, and suggests that aging-related alterations in sensory innervation within the abdominal region may have broader implications for visceral perception and awareness. A recent study has investigated the influence of aging on sensory signaling in the urethra. Specifically, it highlights reduced urethral pressure and afferent activation in older rats compared to young rats in response to fluid flow [25]. These findings suggest that aging significantly affects the sensitivity of urethral afferents. The reported aging-related changes in urethral sensory signaling are in line with the results reported here, and collectively underscore aging-related alterations in afferent activity, suggesting that the sensitivity and density of sensory nerve fibers in the lower urinary tract may decrease with age. Research investigating current perception thresholds upon electrical stimulation of the urethra in women reported increases in perception thresholds associated with aging and overactive bladder [26]. Their findings suggest that deterioration of urethral innervation contributes to aging-associated changes in lower urinary tract function.
The reported negative changes in trigonal sensory innervation in aged rats likely affects CNS processes associated with visceroceptive awareness (e.g., monitoring of bladder volumes and desire to void). Resulting (mal)adaptive processes in the CNS may be contributors to aging-related increases in the prevalence of LUTS. In aging adults, these processes are likely associated with pathological LUT sensations such as those seen in, for example, overactive or underactive bladder or urinary incontinence. A better understanding of aging-related alterations in the bladder sensory web are essential to fully understand these pathologies and improve treatment options for patients. Future research should aim to link peripheral measures of lower urinary tract sensation (such as nerve fiber density and distribution, current perception threshold in the lower urinary tract, and bladder sensations during filling) to CNS processes associated with sensory processing. Neuroimaging studies incorporating manipulation of bladder volumes and assessment of bladder sensations with brain activity and connectivity patterns may help to further understand the impact of aging-associated changes in bladder sensory innervation. Recent neuroimaging developments in the field of neuro-urology highlight the added value of interdisciplinary approaches to improve our understanding of the central control of the lower urinary tract [27-29].
Future studies should further investigate the specific mechanisms underlying the observed age-related alterations in sensory nerve fibers. This could involve examining changes in the expression of neurotrophic factors, receptors, or signaling pathways that are critical for sensory nerve maintenance and function. Identifying the molecular and cellular processes responsible for these changes may offer potential targets for therapeutic interventions aimed at preserving or restoring sensory innervation in aging individuals. Furthermore, given the regional variations in the impact of aging on nerve fibers, as evidenced by regional changes in epidermal innervation [24], it may be beneficial to conduct comprehensive spatial mapping of sensory changes within the lower urinary tract. This could be achieved by assessing sensory innervation in functional and structural sub-regions of the bladder and urethra to determine if specific areas are more susceptible to age-related alterations. Such spatial mapping may provide valuable insights into the localized effects of aging on sensory signaling and help identify specific regions requiring targeted interventions.
To conclude, based on the findings reported here, we argue that aging-associated damage to the sensory system in the trigonal mucosa may be a likely contributor to the development or exacerbation of LUTS. To our knowledge, the detrimental effects of aging on sensory nerve fibers in the bladder trigone has not been previously quantified. Multiple factors could influence the structural and functional integrity of the web of sensory nerves in the trigonal mucosa of aged animals (such as increased oxidative stress in aging [4, 6]), and communication between the periphery and the CNS is likely compromised as a result. Our study elucidates how aging can detrimentally affect morphological and functional characteristics of the PNS, including observable degenerative changes in afferent nerve fibers, which may contribute to a decline in visceroceptive awareness.
NOTES
Grant/Fund Support
This project was funded by NIH/NIA R01 AG056944, R01 DK135076 grant awarded to Prof. Dr. Lori A. Birder.
Research Ethics
The University of Pittsburgh Institutional Animal Care and Use Committee (IACUC) approved all procedures which conform to the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication No. 85-23, revised 1996).
AUTHOR CONTRIBUTION STATEMENT
· Conceptualization: MMDR, GAVK, LAB
· Data curation: MMDR, AWJ
· Formal analysis: MMDR, SP
· Funding acquisition: LAB
· Methodology: MMDR, AWJ, LAB
· Project administration: AWJ, LAB
· Visualization: MMDR, SP
· Writing – original draft: MMDR, SP, LAB
· Writing – review & editing: MMDR, SP, AWJ, JH, GAVK, LAB
REFERENCES
1. Gibson W, Wagg A. Incontinence in the elderly,’normal’ageing, or unaddressed pathology? Nature Rev Urol 2017;14:440-8. 

2. Irwin DE, Milsom I, Hunskaar S, Reilly K, Kopp Z, Herschorn S, et al. Population-based survey of urinary incontinence, overactive bladder, and other lower urinary tract symptoms in five countries: results of the EPIC study. Eur Urol 2006;50:1306-15. PMID: 17049716


3. Rohrmann S, Katzke V, Kaaks R. Prevalence and progression of lower urinary tract symptoms in an aging population. Urology 2016;95:158-63. PMID: 27346671


4. Birder LA, Kullmann AF, Chapple CR. The aging bladder insights from animal models. Asian J Urol 2018;5:135-40. PMID: 29988876


5. Phelps C, Moro C. Ageing and the bladder mucosa: A scoping review of recent animal model studies. Aust N Z J Cont J 2022;28:77-83. 
6. de Rijk MM, Wolf-Johnston A, Kullmann AF, Taiclet S, Kanai AJ, Shiva S, et al. Aging-associated changes in oxidative stress negatively impacts the urinary bladder urothelium. Int Neurourol J 2022;26:111-8. PMID: 35793989




7. Cameron OG. Visceral sensory neuroscience: interoception. Oxford: Oxford University Press; 2001.
8. Craig AD. How do you feel? Interoception: the sense of the physiological condition of the body. Nat Rev Neurosci 2002;3:655-66. PMID: 12154366



9. Khalsa SS, Rudrauf D, Tranel D. Interoceptive awareness declines with age. Psychophysiology 2009;46:1130-6. PMID: 19602175



10. Murphy J, Geary H, Millgate E, Catmur C, Bird G. Direct and indirect effects of age on interoceptive accuracy and awareness across the adult lifespan. Psychon Bull Rev 2018;25:1193-202. PMID: 28685271



12. Purves JT, Spruill L, Rovner E, Borisko E, McCants A, Mugo E, et al. A three dimensional nerve map of human bladder trigone. Neurourol Urodyn 2017;36:1015-9. PMID: 27265789



13. Gabella G, Davis C. Distribution of afferent axons in the bladder of rats. J Neurocytol 1998;27:141-55. PMID: 10640174

14. Andersson KE, McCloskey KD. Lamina propria: the functional center of the bladder? Neurourol Urodyn 2014;33:9-16. PMID: 23847015



15. Kanai A, Andersson KE. Bladder afferent signaling: recent findings. J Urol 2010;183:1288-95. PMID: 20171668



16. de Groat WC, Yoshimura N. Afferent nerve regulation of bladder function in health and disease. Handb Exp Pharmacol 2009;(194):91-138.
17. Suskind AM. The aging overactive bladder: a review of aging-related changes from the brain to the bladder. Curr Bladder Dysfunct Rep 2017;12:42-7. PMID: 28947924




18. Verdú E, Ceballos D, Vilches JJ, Navarro X. Influence of aging on peripheral nerve function and regeneration. J Peripher Nerv Syst 2000;5:191-208. 
19. Mossa AH, Cammisotto PG, Shamout S, Campeau L. Imbalance of nerve growth factor metabolism in aging women with overactive bladder syndrome. World J Urol 2021;39:2055-63. PMID: 32870355



20. Al‐Shawi R, Hafner A, Olson J, Chun S, Raza S, Thrasivoulou C, et al. Neurotoxic and neurotrophic roles of proNGF and the receptor sortilin in the adult and ageing nervous system. Eur J Neurosci 2008;27:2103-14. PMID: 18412630


21. Ochodnicky P, Cruz CD, Yoshimura N, Cruz F. Neurotrophins as regulators of urinary bladder function. Nat Rev Urol 2012;9:628-37. PMID: 23045265



22. Ochodnický P, Cruz CD, Yoshimura N, Michel MC. Nerve growth factor in bladder dysfunction: contributing factor, biomarker, and therapeutic target. Neurourol Urodyn 2011;30:1227-41. PMID: 21520250



23. Birder LA. Is there a role for oxidative stress and mitochondrial dysfunction in age-associated bladder disorders? Ci Ji Yi Xue Za Zhi 2020;32:223-6. PMID: 32955518


24. Lauria G, Holland N, Hauer P, Cornblath DR, Griffin JW, McArthur JC. Epidermal innervation: changes with aging, topographic location, and in sensory neuropathy. J Neurol Sci 1999;164:172-8. PMID: 10402030


25. Geramipour A, Danziger ZC. Age is associated with reduced urethral pressure and afferent activity in rat. Physiol Rep 2021;9:e15107. PMID: 34755496




26. Kenton K, Lowenstein L, Simmons J, Brubaker L. Aging and overactive bladder may be associated with loss of urethral sensation in women. Neurourol Urodyn 2007;26:981-4. PMID: 17626276


27. Clarkson BD, Karim HT, Chermansky CJ, Banihashemi L, Tyagi S, Griffiths DJ, et al. Changes in brain response to urgency before and after treatment of urgency urinary incontinence with onabotulinumtoxin A. Neurourol Urodyn 2022;41:1703-10. PMID: 35904238



28. de Rijk MM, Janssen JM, Chadily SF, Birder LA, Rahnama’i MS, van Koeveringe GA, et al. Between-subject similarity of functional connectivity-based organization of the human periaqueductal gray related to autonomic processing. Front Neurosci 2022;16:1028925. PMID: 36340789


29. Mazeaud C, Salazar B, Braun M, Hossu G, Khavari R. Functional MRI in neuro-urology: a narrative review. Prog Urol 2023;33:349-56. PMID: 37062631


Fig. 1.
(A) Representative image of the trigonal mucosa of young rats. Areas positive for calcitonin gene-related peptide are highlighted in yellow, and nuclei are shown in blue. White arrows indicate fine afferent nerve fiber endings that penetrate deep into the urothelial layers. (B) Representative image of the trigonal mucosa of aged rats. Fine afferent nerve fiber endings are absent from the urothelial layers, and the fibers appear fragmented.
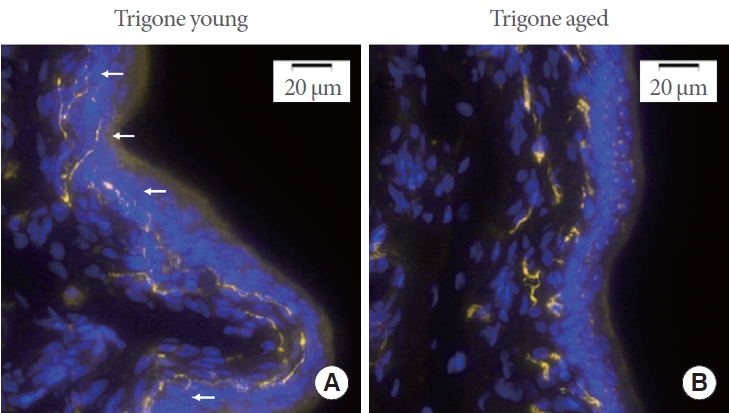
Fig. 2.
(A) Representative binary calcitonin gene-related peptide (CGRP) mask of the trigonal mucosa from young rats. CGRPpositive pixels following region-of-interest detection are displayed in black. (B) Representative binary CGRP mask of the trigonal mucosa from aged rats. (C) Trigonal tissue from aged rats exhibits a significantly reduced area of CGRP positivity (μm2) compared to that of young animals (P=0.0049). Asterisks denote levels of significance: *P≤0.05, **P≤0.01, and ***P≤0.001.
-
METRICS

-
- 0 Crossref
- 1 Scopus
- 1,248 View
- 163 Download
- We recommend




