Modulation of Alpha 1 Adrenergic Receptors on Urinary Bladder in Rat Spinal Cord Injury Model
Article information
Abstract
Purpose
Whereas many studies have focused on the vesical changes of the α1 adrenergic receptor (AR) subtypes in partial outlet obstruction, few studies have addressed the modulation of the α1 AR subtypes after spinal cord injury (SCI). Therefore, we studied the modulation of the α1 ARs in urinary bladder in a rat SCI model.
Methods
Four weeks after a SCI, the whole vesical bodies from eight female Sprague-Dawley rats and from eight controls were harvested. The total RNA was extracted from the samples and was used to prepare cDNA. We developed standard plasmid constructs of glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and three α1 ARs (α1a, α1b, and α1d) to convert the cycle threshold (Ct) values from real-time polymerase chain reaction (RT-PCR) into subtype mRNA concentrations. The detected Ct values of 16 samples from RT-PCR were interpolated into the standard plasmid curves.
Results
All serially diluted standard samples showed very good linearity. The mRNA expression of GAPDH was higher in the SCI group, whereas the mRNA expression of all α1 ARs was lower in the SCI group than in the control animals. The α1a, α1b, and α1d mRNA expression in the controls was 81.7%, 3.3%, and 15.1%, respectively, whereas the α1a, α1b, and α1d mRNA expression in the SCI group was 33.5%, 5.2%, and 60.9%, respectively.
Conclusions
SCI moderates the α1 AR mRNA subtypes in the urinary bladder. The relatively increased α1d or decreased α1a AR mRNA expression may be a therapeutic candidate for controlling the symptoms of neurogenic bladder after SCI.
INTRODUCTION
The prevalence of spinal cord injury (SCI) in young patients is increasing, thus leading to an increased need for lifelong care of these patients [1]. For this reason, it is increasingly important to understand the complications in the urinary bladder of SCI individuals who complain of voiding symptoms or impaired bladder emptying.
Detrusor-sphincter-dyssynergia (DSD), a condition characterized by simultaneous contraction of the detrusor and urethral sphincter, is one of the causes of inefficient bladder emptying after SCI [2].
Traditionally, α1a adrenergic receptor (AR) antagonists have been used to decrease the maximum urethral closing pressure and the symptoms of DSD after SCI [2,3]; however, recent studies have shown that the roles of the α1a ARs extend beyond the urethra or the urinary bladder neck. Because all α1a AR subtypes are expressed in the bladder body, it is reasonable to assume that selective α1 AR antagonists may have on specific roles not only in the bladder neck but also in the bladder body [4-7]. However, the molecular characterization of the AR subtypes in the body of the urinary bladder is not well established.
Three α1 ARs (α1a, α1b, and α1d) have been cloned with the use of molecular technologies and have been characterized pharmacologically [8,9]. Because the α1a AR subtype predominates in the smooth muscle of the prostate and the proximal urethra, this subtype has been assumed to be responsible for the dynamic component of obstruction and the related voiding symptoms [8-12]. Interestingly, the relative expression of the α1 AR subtypes is changed with chronic outlet obstructive lesions in the rat urinary bladder, with a remarkable increase in bladder α1d AR expression but a decrease in bladder α1a AR expression [7]. These findings imply that the α1d AR may be a new therapeutic target for controlling irritable bladder symptoms [12].
The recently developed selective α1d AR antagonist naftopidil and the selective α1a AR antagonist silodosin are used for the treatment of lower urinary tract symptoms around the world. Interestingly, the selective α1d AR antagonist naftopidil improves not only voiding symptoms but also storage symptoms in patients with benign prostatic hyperplasia [13]. The improvement of storage symptoms such as urgency and frequency may be assumed to be from the vesical modulation of α1 AR subtypes in chronic urinary obstructive lesions [7,12,13].
It is well known that ineffective bladder emptying in SCI patients causes the accumulation of residual urine and bladder hypertrophy similar to like the plasticity of the urinary bladder in a chronic urinary obstructive model [1,2]. Even though the phenotypes in the urinary bladder are similar between these two types of dysfunction, it is clear that the SCI model and the chronic urinary obstructive model have different causal mechanisms and different pathophysiologic or molecular responses. Furthermore, whereas the genetic modulations of the α1 AR subtypes in the urinary bladder are relatively well documented in the chronic obstructive model, there are few reports on the changes of subtypes of α1 AR subtypes in detrusor muscle after SCI. Furthermore, which of these selective antagonists will be more clinically useful in the treatment of voiding symptoms after SCI is not established. For these reasons, we studied the subtype changes of the α1 ARs in the urinary bladder in a rat SCI model. Understanding the pharmacologic modulation of the α1 AR subtypes in the rat model of SCI and the mechanisms underlying this modulation may have implications for the management of neurogenic bladder after SCI.
MATERIALS AND METHODS
Animals
All experimental procedures were conducted in accordance with the National Institutes of Health (NIH) guidelines for investigators and those for animal users developed by the NIH animal research advisory committee. A total of 16 Sprague-Dawley female rats, with body weights of 250 g, were used. For SCI, the eighth thoracic cord segment of eight rats was exposed under halothane anesthesia and injury was produced by dropping a 2.0 mm diameter rod with a weight of 10 g from a height of 25 mm onto the exposed spinal cord by use of the New York University Impactor (New York, NY, USA) [14]. Eight controls were subjected to the same operation as the SCI rats but without the contusion injury. Animal care after SCI was performed. The post-injury bladder was manually voided twice daily until voluntary voiding occurred. We assessed the changes in locomotor function after SCI and then maintained the animals for 4 weeks. All animals were terminated at four weeks after SCI. The whole bladders were removed and weighed. The harvested bladder was then stored at -80℃. The control rats were sacrificed at the same time as the SCI group.
mRNA Quantification of α1 AR Subtypes: Real-Time Polymerase Chain Reaction (PCR)
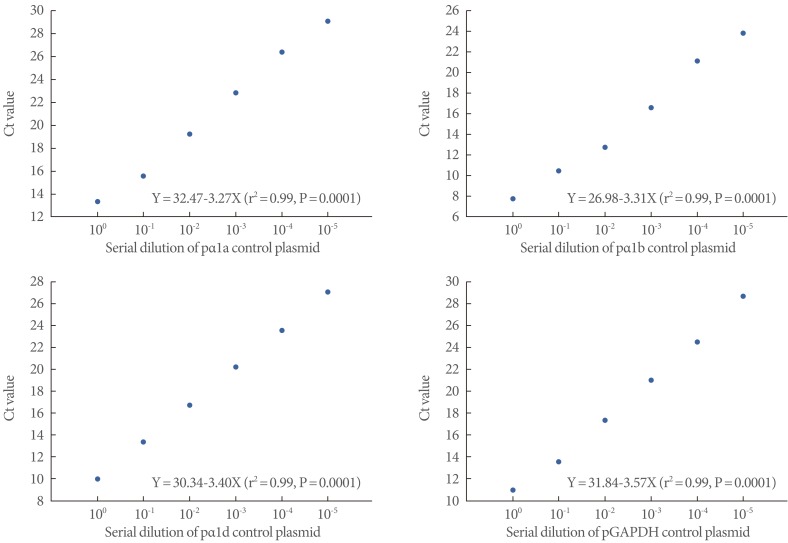
Total RNA was extracted from the whole bladder specimens by using Trizol reagent (Invitrogen, Carlsbad, CA, USA) and chloroform. We designed PCR primers for α1a AR, α1b AR, α1d AR, and glyceraldehyde-3-phosphate dehydrogenase (GAPDH), and all primers spanned at least one intron of the corresponding gene (Table 1). We used the ImProm-II Reverse Transcription System (Promega, Madison, WI, USA) to prepare cDNA according to the manufacturer's instructions. To estimate the mRNA expression, calibration curves were made from the measured fluorescence of dilution series of the control plasmids. Then, the concentrations of the unknowns were calculated from standard values. To obtain constructs for plasmid controls (pα1a AR, pα1b AR, pα1d AR, and pGAPDH) for quantifying the mRNA of the AR subtypes and GAPDH, partial cDNAs were amplified from a rat urinary bladder sample by using each AR subtype and GAPDH primers (Table 1). These cDNAs were then cloned into the pGEM-T Easy vector (Promega) according to the manufacturer's instructions. Finally, the DNA sequences of the cloned plasmids were checked with the restriction enzyme EcoRI (Fig. 1).
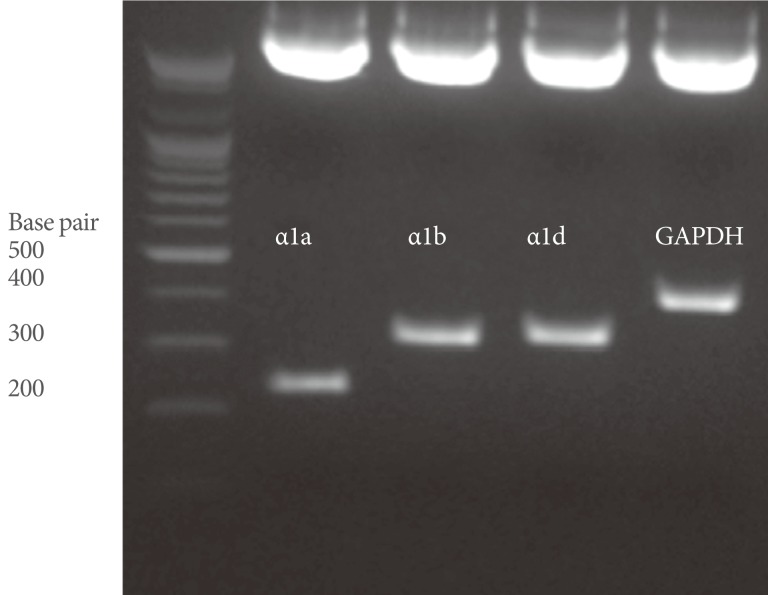
The cloned control plasmids, pα1a AR, pα1b AR, pα1d AR, and pGAPDH were cut with restriction enzyme (EcoRI). We can see the fragmented or restricted segments, measuring about 200- to 400 bp. GAPDH, glyceraldehyde-3-phosphate dehydrogenase.
Two microliters of the standard plasmid was added to 25 µL of reaction mixture containing each forward primer (300 nM), reverse primer (300 nM), 0.5 unit of uracil-N-glycosylase (Sigma Chemicals, Balcatta, WA, USA), and 2X QuantiTech SYBR Green PCR Master Mix (Qiagen GmbH, Hilden, Germany). Real-time PCR was performed in a Rotor-Gene (Rotor Gene 6.0, Corbett, Sydney, Australia) according to the manufacturer's instructions. Amplification consisted of 2 minutes at 50℃ for carryover prevention, 15 minutes at 95℃, 10 seconds denaturation at 95℃, and annealing at 52℃ for 15 seconds and 72℃ for 20 seconds for 40 cycles. Melting curve analysis was performed and concentration values were measured. Negative controls were also included. The detected cycle threshold (Ct) values were interpolated into the standard curves of the plasmid constructs, and the mRNA expression in the samples was calculated [15].
Statistical Analyses
The changed mRNA concentration in each α1 AR subtype of SCI rats was compared with the controls by using the Mann-Whitney test. The statistical difference in the adjusted mRNA expression of the α1 AR subtypes in the controls and the SCI group was determined by Kruskal-Wallis analysis with post-hoc Mann-Whitney test. P-values<0.05 were considered statistically significant.
RESULTS
All rats showed a typical posture of SCI, the hind limbs were drooped and paralyzed at day 1 after the contusion injury. Significant increase in bladder wet weight was noted in the SCI group (Fig. 2).
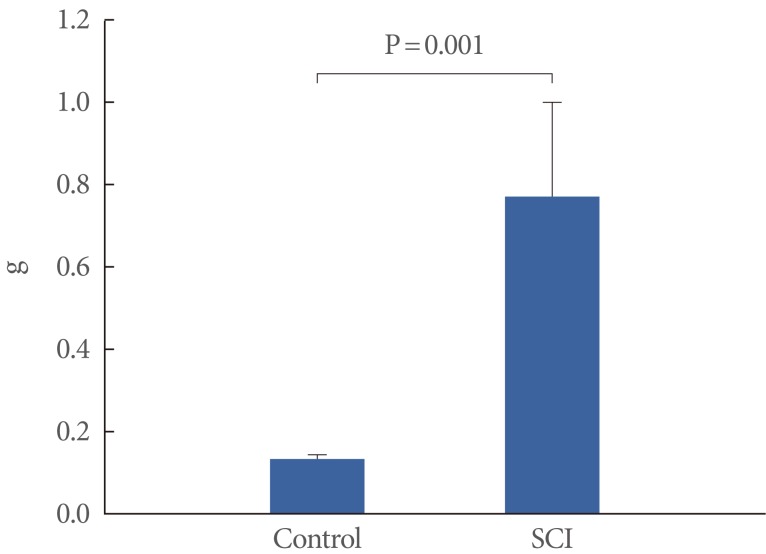
Weight of urinary bladder in the control and the spinal cord injury (SCI) group. Data are reported as the mean±SD.
Standardization of Real-Time PCR
The linear relationships of all cloned plasmids from 100 to 10-5 starting copies/reaction revealed high correlations between the copy numbers and the Ct values. pGAPDH also generated a good log-linear regression plot (Fig. 3).
Changes in mRNA Expression of α1 AR Subtypes
The SCI group showed statistically increased mRNA expression of GAPDH at 4 weeks after injury compared with the control group (Table 2, Fig. 4A) (P=0.041). The expression of the α1 AR subtypes was adjusted by the expression of GAPDH in each sample. The adjusted α1 AR subtype expression in the control group was 371.50±118.19 for α1a, 15.11±7.97 for α1b, and 68.62±40.99 for α1d AR. There were significant differences in AR subtype expression in the urinary bladder in controls (P=0.001) (Fig. 4B).
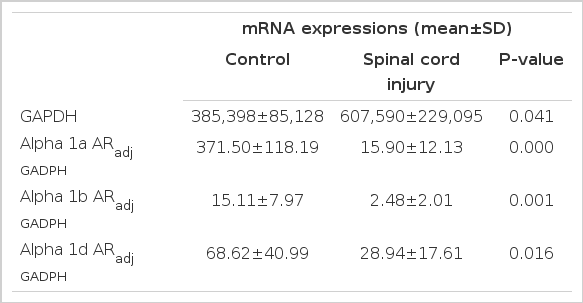
Change in mRNA expression of α1 adrenergic receptor (AR) subtypes in urinary bladder after spinal cord injury
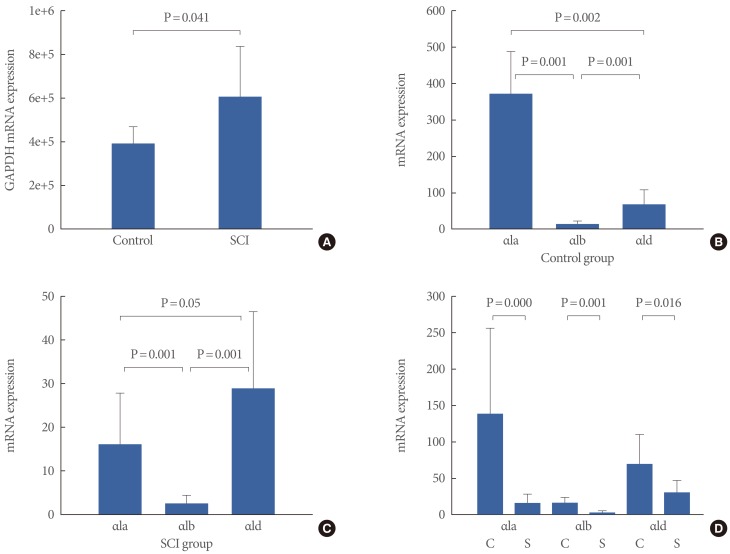
The changes in mRNA expression of α1 adrenergic receptor (AR) subtypes and glyceraldehyde-3-phosphate dehydrogenase (GAPDH). (A) GAPDH mRNA expression in the control and the spinal cord injury (SCI) group. (B) Alpha1 AR subtype mRNA expression in the control group. (C) Alpha1 AR subtype mRNA expression in the SCI group. (D) Expression of each α1 AR subtype between the control and the SCI group. Data are reported as the mean±SD. C, control group; S, SCI group.
The AR subtype expression of the SCI group differed from that of the controls. The adjusted α1 AR expression in the SCI group was 15.90±12.13 for α1a, 2.48±2.01 for α1b, and 28.94±17.61 for α1d AR (Fig. 4C). Furthermore, there were significant differences in AR subtype expression in the urinary bladder in the SCI group (P=0.001). The α1d AR was marginally more expressed than the α1a AR in the SCI group (P=0.05) (Fig. 4C). The SCI group had statistically less mRNA expression of the α1 ARs than did the control group (α1a, P=0.000; α1b, P=0.001; and α1d, P=0.016) (Fig. 4D).
DISCUSSION
The communication between the outlet and the bladder body is under the control of the central nervous system as well as the sympathetic, parasympathetic, and somatic nerves that innervate the bladder body muscle and urinary bladder neck [16,17]. The interplay between the urinary bladder and the upper control organs can be disrupted as a result of trauma or disease [1-3,16,17]. In the case of spinal cord transection, voluntary control of voiding is eliminated and is accompanied by an uncoordinated bladder and external urethral sphincter activity, termed DSD [1-3,16,17]. In addition, a high intravesical pressure can cause damage to the upper urinary tract, leading to vesical muscular hypertrophy [2,18]. Even though many studies have focused on pathophysiological alterations to the bladder neck or posterior urethra after SCI, few studies have been done regarding changes in the enlarged vesical body or hypertrophied detrusor muscle that might accompany SCI [3,4].
Our results showed that the SCI group had statistically increased mRNA expression of GAPDH at 4 weeks after injury compared with the control group (Fig. 4A). GAPDH is one of the most commonly used housekeeping genes for comparing gene expression data [19,20]. The housekeeping genes are standards for normalizing the expression levels of a target gene. Because very few reports have evaluated the GAPDH changes in urinary bladder after SCI, we can extrapolate the significance of the GAPDH change in the urinary bladder after SCI from the findings of the partial bladder neck obstruction model. It is well known that the DNA content and 3H-thymidine incorporation in the urinary bladder are increased in partially obstructed urinary bladders [21,22]. Our data showed that the expression of GAPDH in the SCI group was 2 times that in the controls (Table 2). For these reasons, we adjusted mRNA expression of the α1 AR subtypes to reflect the housekeeping effect of GAPDH.
The α1 AR is a member of the large family of G protein coupled receptors that mediate the actions of the endogenous catecholamines norepinephrine and epinephrine, resulting in smooth muscle contractions in body organs [4-10]. Three subtypes, α1a, α1b, and α1d, have been identified in various mammalian organs and have been characterized by either molecular or pharmacokinetic techniques [4-11]. It is well known that the three α1 AR subtypes expressed in the prostate play a major role in prostatic smooth muscle contraction [11]. The switch on the AR mediates lower urinary tract symptoms and pathophysiologic mechanisms for benign prostatic hyperplasia [23,24]. Quantification of α1 AR mRNA expression within human prostatic tissue has revealed that the α1a AR predominates, followed by the α1d AR and α1b AR [23,24]. Furthermore, the mRNA expression of the α1 AR subtypes in the prostate may be a predictor of the efficacy of subtype selective AR antagonists in patients with benign prostatic hyperplasia [7,23,24].
Because lower urinary tract symptoms or pathophysiological changes in the urinary bladder resulting from bladder outlet obstruction are common disorders in urology, there are ample studies about α1 AR receptor subtype modulation after partial bladder outlet obstruction. For this reason, we compare the α1 AR mRNA modulation in the SCI model with that in the bladder outlet obstruction model induced by partial urethral ligation. Control rats show predominantly α1a AR expression (68 to 74%), followed by α1d AR (21 to 28%) and α1b AR (4 to 5%) along the whole urinary bladder [7]. However, the obstructive rat urinary bladder shows predominantly α1d AR expression (77 to 74%), followed by α1a AR (21 to 24%) and α1b AR (2 to 3%) along the whole urinary bladder [7]. Barendrecht et al. [5] evaluated the mRNA expression of the α1 AR subtypes by use of the real-time PCR method in rats with bladder outlet obstruction induced by partial urethral ligation. In that study, the mean relative expressions (mRNA expression fold of sham) were α1d AR> α1b AR> α1a AR [5]. Interestingly, the mean relative expression of all α1 AR subtypes was higher in the bladder outlet obstruction group than in the sham group; however, discrepancies in subtype expression in urinary bladder were reported by Hampel et al. [7]. In control animals, 70% of α1 AR mRNA was the α1a AR subtype, 5% was α1b AR and 25% was α1d AR, whereas in obstructed bladder, α1 AR expression changed to 23% α1a AR, 2% α1b AR, and 75% α1d AR [7]. In addition, the mRNA expression of the α1a AR subtype was dramatically reduced in the partially obstructed group compared with the control rats [7]. Our results were consistent with the results from Hampel et al.'s report [7] except for one thing. The total expression of α1d AR more increased than in the control animals after 6 weeks of partial outlet obstruction in Hampel et al.'s study [7]. Even though the adjusted α1d AR mRNA expression was dominant among the α1 AR subtypes, we found that the mRNA expression of the α1d AR definitively decreased less than in the control animals. Although the reasons for these differences are not yet clear, the differences may be due to different experimental designs or animal models. Further studies may be needed to elucidate the meaning of the absolute decrease in all α1 AR subtypes in the SCI model.
Because the α1d AR subtype has a 10- to 100-fold higher affinity for the endogenous neurotransmitters norepinephrine and epinephrine than does the α1a or α1b AR subtype, this finding provides a potentially important rationale for targeting the α1d AR subtype when treating voiding symptoms after SCI [7].
To characterize the function of the α1 ARs in the urinary bladder, quantitative evaluation of mRNA and protein must be performed. However, it is methodologically difficult to quantitatively analyze genetic expression by use of western blotting or immunohistochemical staining. Furthermore, because the three α1 ARs have very similar amino acid sequences, it is very difficult to develop unique and specific antibodies for each subtype. Real-time reverse transcriptase PCR is becoming a widely used method to quantify the gene expression from cells, tissues, or tissue biopsies [5,23,24].
A limitation of this study is that we did not evaluate the physiologic role of the changes in α1 AR modulation. Urodynamic studies in animals or contractile responses of harvested strips to selective α1 AR antagonists such as silodosin or naftopidil may extend our findings in the future. In addition, we evaluated α1 AR mRNA expression in whole bladder specimens. Because the α1 ARs are widely distributed throughout the whole urinary bladder, some expression of α1 ARs in the mucosa or submucosa may have affected the mRNA expression values in our experiment.
In conclusion, SCI modulates the α1 AR mRNA subtypes in rat urinary bladder. The relatively increased the α1d or decreased the α1a AR mRNA expression may be a therapeutic candidate for controlling symptoms of neurogenic bladder after SCI. Further studies will be needed to evaluate the action of selective α1 AR subtype antagonists on the urinary bladder after SCI.
ACKNOWLEDGEMENTS
This study was financially supported by Institute of Medical Science Research of Dankook University Medical Center in 2010.
Notes
No potential conflict of interest relevant to this article was reported.