INTRODUCTION
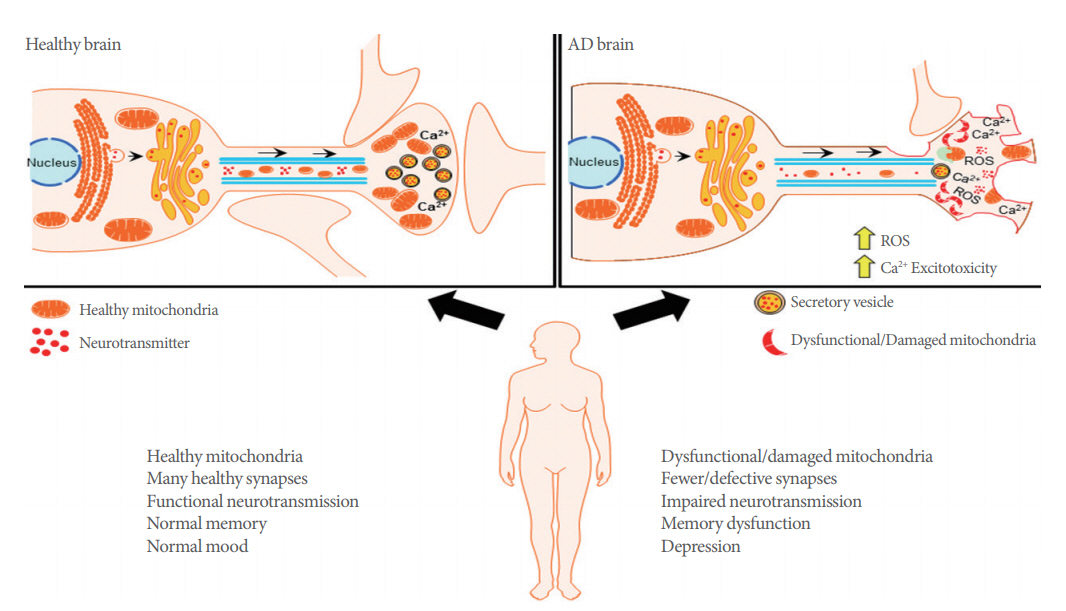
Alzheimer disease (AD) is an aging-associated neurodegenerative disorder that is clinically characterized by progressive cognitive dysfunction and eventual death. The currently established main pathological features of this devastating disease include extracellular Amyloid-beta (Aβ) plaque deposits and intracellular neurofibrillary tangles (NFTs) [1]. Although these pathological features have undergone decades of intensive basic science and clinical investigations for potential application of preclinical and clinical therapeutic strategies it remains unclear whether these cellular and molecular phenotypes drive AD pathogenesis. Recently, mitochondria have gained accepted status as a key player contributing to normal aging and AD pathogenesis [2]. Investigations of this essential organelle in AD suggest that mitochondria dysfunction and oxidative stress are early contributory events to AD progression, thus making mitochondria an appealing therapeutic target. Supporting this hypothesis, brain imaging studies have demonstrated defects in glucose metabolism in AD patients, an abnormality that may occur well before the onset of clinical symptoms [3]. Interestingly, mitochondrial abnormalities have been reported as an early and prominent feature in AD patient brains [4] and AD transgenic mouse models [5,6].
Mitochondria are double-membraned organelles responsible for large amounts of cellular energy production derived from the biochemical processes of respiration. It provides most of the cellular adenosine triphosphate (ATP) demand by oxidative phosphorylation. Unfortunately, the high demand for energy that drives ATP mediated cellular functions, also generates a significant amount of reactive oxygen species (ROS) as a byproduct of ATP biogenesis [7]. These organelles also critically regulate other essential cellular functions including fatty acid oxidation, glutamate and urea metabolism, as well as cell death via activation of cellular apoptosis [8]. Notably, several reports indicate that AD-patient derived mitochondria are differentially affected in comparison to age-matched patients without dementia [9]. These disturbances include impaired oxidative phosphorylation, energy metabolism, as well as excess generation of ROS, altered mitochondrial biogenesis and transport (Fig. 1) [10]. Not surprisingly, in areas highly associated with cognitive function, such as the hippocampus and cortex, there is evidence for high levels of impaired mitochondrial respiration, excessive ROS production, as well as aberrant disruptions in membrane potential, and cytochrome c oxidase activity. Interestingly, brain structures that are less involved in complex cognitive ability, such as the striatum or amygdala, mitochondria-regulated processes are minimally to moderately affected [11]. In this review, we will report on the association between mitochondrial dysfunction and synaptic deficits in AD, which we believe underlies critical neuropathology of this neurodegenerative disease. We will also discuss how protection of synaptic transmission by targeting mitochondrial bioenergetics processes can provide an efficacious approach toward our understanding of mitochondrial mechanisms of neurodegeneration. More importantly, this review will provide a basis for future therapeutic strategies that will advance our understanding of AD and provide novel treatment approaches for this devastating disease.
MITOCHONDRIAL TRAFFICKING AND SYNAPTIC FUNCTION
Mitochondria derived ATP is involved in the complex regulation of synaptic plasticity and maintains the essential functions at synapses. For example, ATP generated from oxidative phosphorylation is integral for maintenance of the ion gradients required for axonal and synaptic membrane potentials [12]; mobilizing synaptic vesicles from reserve pools to release sites [13]; supporting vesicular neurotransmitter release, neurotransmitter recycling [14]; and supporting dynamic synaptic assembly and plasticity [15]. Neural presynaptic and postsynaptic terminals have high demands for ATP due to the extensive energy cost required to maintain homeostatic calcium flux during neurotransmission, and this persistent energy expenditure requires the constant presence of mitochondria [15], where they help to maintain neurotransmission by producing ATP and buffering Ca2+ at synapses [16]. Compared with nonsynaptic mitochondria, synaptic mitochondria showed a greater degree of age-dependent accumulation of Aβ, increased mitochondrial oxidative stress and impaired respiration in an AD mouse model [17]. The importance of adequate synapse function is evident in neurodegenerative disease, given that loss of synapses is one of the early pathological features of AD, and a strong correlation exists between the extent of synapse loss and the severity of dementia (Fig. 1) [18,19].
While mitochondrial biogenesis plays an essential role in homeostatic maintenance of synaptic function, efficient mitochondrial mobility is important in enabling the rapid distribution of this organelle to different areas in order to meet the high metabolic demands of neurons [20]. Motile mitochondria can become stationary but can be remobilized and redistributed in response to changes in cellular metabolic status and synaptic activity [21]. Dual-channel imaging of neuronal mitochondria in hippocampal synaptic terminals indicate several transitional motility patterns of axonal mitochondria. These include stationary mitochondria positioned outside of synapses or others that are docked at synapses. Other patterns are described as motile mitochondria that are either passing through synapses, or those that are paused at synapses for short or long time periods before re-engaging movement [22]. Mitochondrial trafficking is known to be impaired in AD and this transport defect is thought to contribute to the pathogenesis of neurodegeneration. For example, the absence of mitochondria in presynaptic terminals may reduce local ATP supplies which affect ATP-dependent vesicular neurotransmitter loading or reduce the pool of ATP available for working the myosin motors required to transport synaptic vesicles. Similarly, deficiencies in ATP can reduce the available energy to power the Na+/Ca2+ exchange process which is important for removing Ca2+ from nerve terminals, thus setting up a scenario that drives Ca2+ derived cellular excitotoxicity [23,24]. Interestingly, there are reports which suggest that Aβ and intracellular NFT are potential driving forces behind mitochondrial trafficking deficits in AD pathology. A study by Zhao et al. [25] revealed that drastic reductions in the number of axonal mitochondria was associated with a significant increase in mitochondrial size, thus suggesting that mitochondrial trafficking and mitochondrial fusion-fission are intertwined mechanisms.
Although motile mitochondria perform these essential functions, stationary docked mitochondria are also required to meet the high energy demand of axonal and synaptic functions. Docked mitochondria serve as stationary power plants for stable and continuous ATP supply necessary to maintain the activity of Na+/K+ ATPase, fast spike action potential propagation, and synaptic transmission [21]. Synaptic membrane proteins traditionally known to be involved in inhibition of SNARE vesicular neurotransmitter docking and release, have also been shown to play a role in mitochondrial anchoring. One such protein, Syntaphilin, has been identified to be the mitochondrial-anchoring protein that specifically immobilizes this organelle in axons [16]. Dysfunctional mitochondria manifest retrograde transport toward the soma, and this maladaptive behavior is typified by the selective release of syntaphilin which results in subsequent activation of PARK2/Parkin-mediated mitophagy. Notably, this syntaphilin-mediated response is robustly activated during the early disease stages of AD-related cortical neuron dysfunction [26,27].
MITOCHONDRIAL CA2+ HEMOSTASIS AND SYNAPTIC PLASTICITY
Besides supplying ATP, mitochondria also buffer Ca2+ in neuronal presynaptic terminals and dendrites [28]. The mitochondrial membrane utilizes its membrane potential to initiate and maintain Ca2+ ion flux in the mitochondrial matrix. The reversibility of this phenomenon is essential for Ca2+ storage and release in brain mitochondria [29]. Moreover, balanced Ca2+ levels in the mitochondrial matrix tightly regulate oxidative phosphorylation activity, thus adequately maintaining the rate of ATP production [30]. However, if the levels of Ca2+ accumulation overwhelm the ability for removal of Ca2+ within mitochondria, it can result in detrimental cytotoxicity. This phenomenon is often a typical byproduct of glutamate hyper-excitation during excessive excitatory neurotransmission and results in cellular excitotoxicity [29]. The ability for mitochondrial Ca2+ buffering is a critical function that must be maintained by this organelle as aberrant Ca2+ sequestration causes increased production of mitochondrial ROS. Therefore, for adequate synaptic function and plasticity, a fine balance must be achieved since ROS donors produce long-term potentiation (LTP), while conversely, ROS scavengers have been reported to block LTP [31]. Increased mitochondrial superoxide ROS can activate Ca2+-dependent protein kinases that are critical for synaptic plasticity [32]. Excessive Ca2+ uptake into mitochondria often leads to mitochondrial membrane permeabilization and induction of apoptosis. Also, Ca2+ dysregulation occurring in other cell types that support neuronal activity may contribute to degenerative neuropathology [33]. Therefore, the intracellular homeostasis of Ca2+ in neuronal mitochondria plays a significant role in energy production and maintenance of synaptic plasticity. The imbalance of Ca2+ within neuronal mitochondria is known to play an important role in the onset of AD. Modulation of mitochondrial Ca2+ is a potentially promising pharmacological therapeutic target for future AD treatment.
MITOCHONDRIA DNA METHYLATION
In humans, mitochondrial DNA (mtDNA) has approximately 37 genes, all of which are essential for normal mitochondrial function. DNA methylation is a well-known epigenetic mechanism that regulates nuclear gene transcription. Early studies showed that AD brains possessed consistent global reductions in DNA modifications of 5-methylcytosine (5mC) and 5-hydroxymethylcytosine [34]. Given the high heritability estimates for AD, there is additional supporting evidence suggest that epigenetic-mediated reductions in the expression of genes that form complex I-and-IV subunits of the electron transport chain (for example ND4), are particularly prevalent in AD brains [35,36]. Furthermore, genetic studies have identified somatic mutations in several mitochondrial encoded cytochrome-c oxidase genes, as well as in the early mitochondrial replication intermediate displacement loop (D-loop), the mutations which probably alters mitochondrial transcription, replication, and mtDNA deletions in AD brains [37,38].
In recent human studies, the mitochondrial 5mC levels were measured from the entorhinal cortex in AD patients and control brains, along with an APP/PS1 transgenic mouse model at 3, 6, and 12 months of age. The authors report the presence of mitochondrial 5mC in CpG and non-CpG sites in the hippocampal entorhinal cortex. Moreover, compared to control cases, AD patients had increased expression of 5-mC found in the Dloop region of mtDNA in the entorhinal cortex. In their APP/PS1 mouse model, they observed a dynamic pattern in the content of mitochondrial 5mC, as AD pathology progressed across 3, 6, and 12 months of age. This suggests that mtDNA epigenetic modulation in human brain is vulnerable to neurodegenerative disease states [39]. The methylation levels of the mtDNA D-loop region detected in blood DNA from 133 AD patients and 130 controls had observed a significant 25% reduction of DNA methylation in AD patients [40]. Further studies showed that when targeting, mtDNA, methylating enzymes modify cytosine in the CpG or GpC context. Interestingly, mtDNA gene expression remained unchanged upon induction of CpG methylation, whereas induction of C-methylation in the GpC context decreased mtDNA gene expression [41].
Given the important role of mitochondria in AD, exploring the impact of mtDNA methylation will add to our understanding of the role of mitochondria in AD pathogenesis. Numerous mitochondrial haplogroups and single nucleotide polymorphisms have been reported to influence risk for AD, but the majority of these studies have not been replicated, nor experimentally validated. It remains challenging to define the relationship between the mitochondrial genome and AD including small numbers of patient subjects, insufficient genetic data, and technical challenges in data analysis [42]. We suggest that with bigger sample sizes and the application of whole genome sequence data, there can be substantially beneficial advancements for the study of mitochondrial genetic variation in AD.
CONCLUSIONS
Mitochondrial dysfunction is an early pathological feature of AD development, which greatly contributes to maladaptive synaptic deficits and loss of synapses in the earlier phase of this disease. Several therapeutic studies have been conducted with promising experimental results, and these studies have identified mitochondria specific ROS scavengers, which will enable a greater concentration of antioxidants to accumulate in mitochondria and allow a more specific method for combatting mitochondrial oxidative stress. The neuroprotective role of a number of currently available agents that modulate mitochondrial Ca2+ transport pathways and buffering capacity are currently under intense investigation by the scientific community. Further, both generic and mitochondria-targeted studies should be carried out to find additional potential treatment strategies for synaptic defects and neuronal degeneration in AD.