 |
 |


- Search
| Int Neurourol J > Volume 27(1); 2023 > Article |
|
ABSTRACT
Purpose
We investigated the effectiveness of intravesical botulinum toxin-A (BTX-A) injection therapy in patients with lower urinary tract dysfunction (LUTD) and upper urinary tract (UUT) deterioration and evaluated whether BTX-A injection therapy could substitute for augmentation uretero-enterocystoplasty (AUEC).
Methods
Data from a prospective, single-center cohort from 2017–2021 were analyzed. Patients were divided into 2 treatment groups: AUEC and BTX-A (i.e., patients who declined AUEC). Bladder and UUT functions were assessed by comparing clinical information, urodynamic data, laboratory results, and imaging records.
Results
In total, 121 patients were enrolled (BTX-A group: 41 patients; AUEC group: 80 patients). The BTX-A group showed a reduced maximum detrusor pressure and increases in the maximum bladder volume and bladder compliance (P<0.05). However, in follow-up evaluations, significantly smaller improvements (all P<0.05) in urodynamic parameters were found in the BTX-A group than in the AUEC group. Notably, there was no significant improvement in vesicoureteral reflux (VUR; P=0.66) or upper urinary tract dilatation (UUTD; P=0.75) in the BTX-A group, and no statistically significant difference in serum creatinine (Scr) levels or the estimated glomerular filtration rate (eGFR) was observed in the follow-up evaluations (all P>0.05). Both VUR and UUTD improved significantly in the AUEC group, and the Scr and eGFR levels significantly improved after AUEC relative to baseline levels (P<0.05). The reduction in the Scr level was significantly lower in the BTX-A group than in the AUEC group during 0–15 months of follow-up (Scr reduction differences, −1.36; P<0.01).
Conclusions
Although BTX-A injection therapy was effective for improving bladder function, BTX-A injections did not alleviate UUT deterioration in this study, particularly in patients with advanced-stage LUTD. Conversely, AUEC for LUTD has a well-established role in improving UUT function. Hence, BTX-A injection therapy should not replace AUEC to ameliorate UUT impairment and protect UUT function.
Patients with refractory lower urinary tract dysfunction (LUTD) caused by neurologic disorders exhibit upper urinary tract (UUT) deterioration involving upper urinary tract dilatation (UUTD) and serum creatinine (Scr) abnormalities due to lower bladder capacity and/or excessive intravesical pressure [1,2]. Augmentation uretero-enterocystoplasty (AUEC), a type of augmentation cystoplasty (AC) performed simultaneously with ureteroplasty and antireflux reimplantation, is an option for patients with UUT deterioration [3]. However, this procedure can involve complications [4], such as metabolic disturbances, urolithiasis, and even malignancy.
Botulinum toxin-A (BTX-A), produced from Clostridium botulinum, inhibits the release of acetylcholine, thereby suppressing contraction of the detrusor muscle [5]. Intravesical BTX-A injection therapy has been recommended as a second-line treatment for patients with neurogenic detrusor overactivity (NDO) [6]. Additionally, BTX-A detrusor injections increase bladder capacity, maintain low detrusor storage pressures, and control urinary incontinence [7]. Hence, the clinical application of BTX-A injection therapy can be further extended to the treatment of refractory LUTD given the following advantages: simple administration, minimal invasiveness, and lack of adverse events.
Currently, given the wide application of BTX-A injections, the number of patients choosing AUEC has significantly declined. However, little research has been conducted to evaluate the protective effect of BTX-A injections in patients with LUTD and UUT deterioration. The purpose of this cohort study was to determine the efficacy of BTX-A injection therapy in patients with UUT deterioration who had declined AUEC and to ascertain whether intravesical BTX-A injection therapy could serve as an alternative treatment to AUEC.
This prospective comparative study was approved by the Ethics Committee of the China Rehabilitation Research Center (No. 2017-003-1). All patients with UUT deterioration who underwent AUEC or declined AUEC to receive BTX-A injections were included between January 2017 and December 2021.
All patient demographics and perioperative results were prospectively collected in our department. The patients who underwent intravesical BTX-A (Hengli; Lanzhou Biological Products, Lanzhou, China) injection therapy received serial 300-U injections at baseline and every 3–6 months thereafter. Patients were permitted to discontinue treatment if they were dissatisfied with the outcome or experienced intolerable adverse events. The AUEC procedures were performed by urological surgeons led by Professor Liao. All patients were treated with anticholinergics postoperatively and were evaluated every 3–6 months for therapeutic effects.
The indications for AUEC have been described in our previous studies [8–10]. The criteria for BTX-A injection therapy included patients who met the criteria for AUEC but declined AUEC. Urodynamic parameters, including maximum bladder capacity (MBC), maximum detrusor pressure (MDP), bladder compliance (BC), and vesicoureteral reflux (VUR), were assessed by video-urodynamic studies (VUDSs), and UUTD was evaluated using a magnetic resonance urography (MRU)-UUTD system [11,12]. Ultrasound does not reveal the kidney and ureter in the same image; thus, the MRU-UUTD system complemented the Society for Fetal Urology grading system in the identification of the entire urinary tract. Patients who declined to complete the follow-up or had previously undergone vesicoureteral reconstruction were excluded.
All statistical analyses were performed using Empower R ( www.mpowerstats.com; X&Y Solutions, Inc., Boston, MA, USA). Continuous variables were expressed as the mean±standard deviation or median (interquartile range [IQR]) and evaluated using the Student t-test, 1-way analysis of variance, the Mann-Whitney U-test, or the Kruskal-Wallis H test. Categorical variables were presented as numbers (percentages) and evaluated with the chi-square or Fisher exact test. All statistical tests were 2-sided, with α=0.05.
General additive mixed models (GAMMs) with smooth curve fitting were used for analyzing repeated measurement data. A GAMM was used to assess the relationships between follow-up duration and MBC, MDP, BC, and Scr level, stratified by the 2 treatment protocols.
In total, 121 consecutive patients were eligible for inclusion in the study, including 41 patients who received intravesical BTX-A injection therapy and 80 patients who underwent AUEC. The median follow-up duration was 17.0 months (IQR, 8.3–21.0 months) in the BTX-A group and 24.0 months (IQR, 6.50–26.00 months) in the AUEC group (P=0.21). The patient demographic characteristics are summarized in Table 1. Compared with patients who underwent AUEC, patients who received BTX-A injection therapy had a higher baseline MBC (152.0 mL vs. 86.0 mL, P<0.01) and were less willing to receive an invasive and irreversible procedure than the AUEC group.
The VUDS outcomes in both groups showed a therapeutic effect on bladder function (Fig. 1). The continuous changes in urodynamic parameters seen in the follow-up evaluations of patients who received serial BTX-A injections are shown in Fig. 2A. Significant improvements in the MBC and BC were noted in the BTX-A group at the initial visit and every 3 months thereafter (all P<0.05). A declining trend in MDP was also noted 1–3 months after treatment compared to the pretreatment level, with a statistically significant difference after 3 months (P<0.05). Similarly, the MBC, MDP, and BC were significantly ameliorated by AUEC 1–3 months after surgery (P<0.05), and the improvements were sustained for >2 years (all P<0.05, Fig. 2B).
The GAMM with smooth curve fitting analyses showed differences in VUDS parameters between the BTX-A and AUEC groups during the follow-up period (Fig. 2C, D). The adjusted increase in MBC for the BTX-A and AUEC groups over 15 months was 13.62 and 22.67 (P<0.01), respectively, as shown in Table 2. A significant difference in the increased MBC was observed between the 2 groups (13.20, P<0.01). Similarly, the improvement in the BC in patients undergoing AUEC was greater 0–15 months after surgery than in the BTX-A injection group (2.82 vs. 1.09, P<0.01). Furthermore, the MBC and BC indices remained stable for patients in both groups at the 15-month follow-up. Interestingly, the MDP levels in both groups exhibited a time-dependent decrease (Fig. 2E), and a difference in the MDP reduction was seen between the BTX-A and AUEC groups throughout the follow-up period (−1.09, P<0.01).
In the BTX-A group, a total of 82 ureters were analyzed. VUR was noted in 20 ureters, including 7 ureters with grade I–II VUR and 13 with grade III–V VUR. At the last follow-up evaluation, the VUR grade showed no improvement compared to baseline and the high-grade (≥III) VUR rate was unchanged (15.85% vs. 13.41%, P=0.66). The specific follow-up information is shown in Fig. 3A. In addition, UUTD was measured in 78 of 82 ureters in the BTX-A group. No improvements in UUTD were found when compared with baseline (Fig. 3B), and no reduction in the number of ureters with high-grade (≥III) UUTD was observed (53.66% vs. 51.22%, P=0.75). Eighty patients and 154 ureters were enrolled in the AUEC group, and there were 71 ureters (46.10%) with persistent VUR. A significant improvement in VUR was detected in the AUEC patients, and 63 of the 71 ureters (88.73%) with VUR resolved after AUEC treatment (Fig. 3C). A quantitative improvement in the MRU-UUTD grades was observed at the last follow-up (Fig. 3D); the rate of high-grade MRU-UUTD decreased from 61.04% to 18.84% (P<0.01).
Renal function, represented by Scr levels, was analyzed as an important index of UUT deterioration, and the evolution of Scr levels over time was recorded for the 2 groups (Fig. 4A, B). Notably, no significant preoperative or postoperative difference was observed in the Scr levels of the BTX-A group (all P>0.05). The Scr level in patients who received AUEC significantly improved 3–6 months after surgery (110.36 μmol/L vs. 86.97 μmol/L, P<0.05), and this improvement was subsequently sustained. Reductions in the Scr level of the 2 groups were significantly different within the 15-month follow-up period (−1.36, P<0.01) (Table 2). The estimated glomerular filtration rates (eGFRs) calculated using the abbreviated Modification of Diet in Renal Disease formula, were opposite to the trend in Scr levels (Fig. 4D, E). Adjusted smooth curve fitting showed that the Scr and eGFR levels displayed marked improvements over time after patients underwent AUEC and remained stable during the postoperative follow-up (Fig. 4C, F). Conversely, in the BTX-A treatment group, no significant change was recorded in the Scr and eGFR levels over time compared with baseline, indicating stable renal function.
Some common complications occurred in the patients undergoing AUEC, including bowel dysfunction in 2 patients (Clavien-Dindo [CD] classification grade II), metabolic acidosis in 8 patients (CD grade II), and bladder calculus in 3 patients (CD grade IIIa); all problems were resolved by appropriate therapy. No apparent side effects or complications were observed in the BTX-A group during the follow-up evaluations. Finally, 11 patients with ineffective BTX-A outcomes were converted to AUEC for permanent correction.
Bladder management for neurogenic UUT deterioration should meet 3 main objectives: low-pressure urine storage, adequate urine drainage, and preservation of the UUT. AC is the mainstay for managing patients with LUTD in whom conservative measures have failed [13]. Previous studies have demonstrated that AC is a safe and effective procedure that can improve bladder function and quality of life parameters, thereby preserving renal function [9,10,14].
Compared with AC, AUEC is better able to protect UUT function for patients with VUR and UUTD with concomitant ureteral stenosis and tortuosity [3]. Because AC/AUEC is an open abdominal procedure involving the digestive and urinary systems, various complications may occur. The wide-ranging application of minimally invasive techniques such as intravesical BTX-A injection therapy in the management of LUTD has led to a sharp decline in the application of AC/AUEC [15]. Previous studies have shown that intradetrusor BTX-A injections improve urgency incontinence episodes and quality of life [5,16]. Theoretically, the detrusor injection of BTX-A improves urodynamic parameters by inhibiting the efferent and afferent pathways [2], which has been demonstrated in cases of poor BC and NDO [17,18]. We compared the therapeutic effects of BTX-A injection therapy and AUEC for patients with UUT impairment. Our results showed that the VUDS parameters after BTX-A injections improved significantly, and were consistent with the results of previous studies [19,20]. The intravesical filling pressure decreased significantly, suggesting that BTX-A injection therapy can establish a compliant bladder with reduced bladder pressure and increased bladder capacity to avoid renal compromise and UUT deterioration. However, because BTX-A produces a temporary (3–6 months) and reversible denervation [21], the potency of the muscle relaxant effect on the detrusor muscle is inadequate and short-lived. Although the smoothing analysis curves for the VUDS parameters revealed that both treatments had a profound impact on improving bladder function, those who underwent AUEC had a significantly greater improvement in MBC, MDP, and BC than patients who received BTX-A injection therapy.
In addition to improved bladder function, improvements in VUR and UUTD are important indicators in the evaluation of both treatment regimens. The protective role of AC/AUEC for UUT in LUTD patients has been recognized [3,8–10]. Experts have argued that intradetrusor BTX-A injections not only make the bladder compliant and help avoid renal compromise and UUTD [22], but have also resulted in significant improvements in VUR [23] and decreased the risk of UUT damage [24]. Our data revealed that, although BTX-A injection therapy can improve bladder function, decrease VUR and UUTD, and prevent renal function deterioration, it remained unsatisfactory and the MRU-UUTD grade distribution was not substantially altered in the last follow-up evaluation. Therefore, this treatment demonstrated little improvement in the renal function of patients with UUT deterioration. We propose that the following factors may account for the unsatisfactory therapeutic effect: First, the urodynamic parameters of BTX-A injection therapy showed less favorable outcomes than AUEC, and the improvements in bladder capacity and pressure were insufficient (maintained around 20 cm H2O) after BTX-A injection therapy. Therefore, we concluded that BTX-A injections only eliminated high-pressure VUR and had little effect on low-pressure VUR (Fig. 1A, B) and UUTD (Fig. 5A, B). Second, BTX-A injections minimally affect detrusor muscle relaxation and have a short duration of action, which impairs the ability to establish an elastic low-pressure reservoir to relieve UUTD. In addition, UUTD is often found with concomitant ureteral stenosis and tortuosity, particularly in patients with advanced-stage neurogenic bladder and with a longer disease duration. BTX-A injection therapy could not correct ureteral stenosis/obstruction and tortuosity above the bladder. Third, because medical developments are uneven across China, bladder wall fibrosis induced by a long history of illness and inappropriate bladder management can disrupt antireflux mechanisms in the ureterovesical junction. Thus, BTX-A injection therapy ameliorates bladder capacity but cannot reconstruct the antireflux mechanism. In contrast, the antireflux reconstruction and ameliorated bladder parameters achieved by AUEC dramatically reduced the postoperative occurrence of VUR (Fig. 1C, D). Importantly, ureteral obstruction and tortuosity can be completely relieved by AUEC through ureterolysis and tailoring to release adhesive bands, straighten tortuous positions, and shorten superfluous ureters. This method ensured unobstructed drainage of urine from the kidneys and improved renal function in patients with LUTD (Fig. 5C, D) [3].
Although BTX-A injection therapy is a less invasive and lower-cost option [25], the treatment requires repeated injections at 6-month intervals to maintain the initial favorable therapeutic effect. In this study, patient follow-up and adherence to treatment were not as high as we had expected. Moreover, the quality of life in the AUEC group was superior to that of the BTX-A group [26], possibly because of the long-term effectiveness of AUEC when compared to intermittent BTX-A injection therapy. Notably, the cost-effectiveness of AUEC gradually emerged as the cost of BTX-A injections increased and the complication rate of AUEC decreased [25]. Ultimately, 11 patients with unsatisfactory outcomes following BTX-A injections were converted to permanent correction with AUEC.
This was the first study to explore the protective effects of BTX-A injection therapy on the UUT and compare the outcomes to AUEC outcomes. In addition, we were the first to objectively report that BTX-A injection therapy failed to alleviate UUT impairment and protect UUT function in all patients with LUTD, especially those with ureteral tortuosity and obstruction that could be improved by AUEC. The main limitation of this prospective, single-center study was the lack of randomization between the 2 patient groups. Due to technology and cost limitations, most patients did not undergo an isotope renogram and cystatin C to evaluate differences in renal function, and Scr levels did not fully reflect renal function. Finally, data from the present comparative study should be validated externally, preferably by multicenter experiences.
In conclusion, minimally invasive intradetrusor BTX-A injection therapy was used to manage LUTD, and was shown to provide urodynamic benefits, including increased bladder capacity and decreased storage pressure. However, therapeutic relief of VUR and UUTD, and improved renal function remained unsatisfactory, particularly in patients with advanced LUTD and longer disease duration. Our data confirmed that AUEC generated greater improvement in urodynamic parameters and UUT function than serial BTX-A injections. Therefore, we believe that intravesical BTX-A injection therapy should not serve as an alternative protocol for AUEC. Although the application of AUEC has been declining in the treatment of LUTD, it still plays an important role for patients with refractory, advanced-stage LUTD.
NOTES
Funding/Grant Support
This study was supported by grants from the National Key R&D Program of China (2018YFC2002203).
REFERENCES
1. Ginsberg DA, Boone TB, Cameron AP, Gousse A, Kaufman MR, Keays E, et al. The AUA/SUFU guideline on adult neurogenic lower urinary tract dysfunction: diagnosis and evaluation. J Urol 2021;206:1097-105. PMID: 34495687


2. Jiang YH, Chen SF, Kuo HC. Frontiers in the clinical applications of botulinum toxin A as treatment for neurogenic lower urinary tract dysfunction. Int Neurourol J 2020;24:301-12. PMID: 33401351




3. Ying X, Liao L. Augmentation uretero-enterocystoplasty for refractory urinary tract dysfunction: a long-term retrospective study. BMC Urol 2021;21:166. PMID: 34847903




4. Wang X, Zhang F, Liao L. Current applications and future directions of bioengineering approaches for bladder augmentation and reconstruction. Front Surg 2021;8:664404. PMID: 34222316



5. Liao L, Liu Q, Cong H, Xu Z, Li E, Weng Z, et al. Hengli((R)) Chinese botulinum toxin type A for treatment of patients with overactive bladder: a multicenter, prospective, randomized, double-blind, placebo-controlled trial. Front Pharmacol 2022;13:840695. PMID: 35250586


6. Michel F, Ciceron C, Bernuz B, Boissier R, Gaillet S, Even A, et al. Botulinum toxin type A injection after failure of augmentation enterocystoplasty performed for neurogenic detrusor overactivity: preliminary results of a salvage strategy. The ENTEROTOX Study. Urology 2019;129:43-7. PMID: 30926380


7. Cruz F, Herschorn S, Aliotta P, Brin M, Thompson C, Lam W, et al. Efficacy and safety of onabotulinumtoxinA in patients with urinary incontinence due to neurogenic detrusor overactivity: a randomised, double-blind, placebo-controlled trial. Eur Urol 2011;60:742-50. PMID: 21798658


8. Zhang F, Liao L. Sigmoidocolocystoplasty with ureteral reimplantation for treatment of neurogenic bladder. Urology 2012;80:440-5. PMID: 22857763


9. Wang Z, Liao L. Effectiveness and complications of augmentation cystoplasty with or without nonrefluxing ureteral reimplantation in patients with bladder dysfunction: a single center 11-year experience. J Urol 2018;199:200-5. PMID: 28743527


10. Liao L, Zhang F, Chen G. Midterm outcomes of protection for upper urinary tract function by augmentation enterocystoplasty in patients with neurogenic bladder. Int Urol Nephrol 2014;46:2117-25. PMID: 25053014



11. Liao L. A new comprehensive classification system for both lower and upper urinary tract dysfunction in patients with neurogenic bladder. Urol Int 2015;94:244-8. PMID: 25115367



12. Liao L, Zhang F, Chen G. New grading system for upper urinary tract dilation using magnetic resonance urography in patients with neurogenic bladder. BMC Urol 2014;14:38. PMID: 24885460




13. Martinez L, Tubre R, Roberts R, Boone T, Griebling TL, Padmanabhan P, et al. Refractory bladder dysfunction: a multi-institutional experience with intravesical botulinum toxin-a injection in adult patients who underwent previous augmentation cystoplasty. Turk J Urol 2020;46:309-13. PMID: 32525475



14. Gurung PM, Attar KH, Abdul-Rahman A, Morris T, Hamid R, Shah PJ. Long-term outcomes of augmentation ileocystoplasty in patients with spinal cord injury: a minimum of 10 years of follow-up. BJU Int 2012;109:1236-42. PMID: 21851549


15. Shreck E, Gioia K, Lucioni A. Indications for augmentation cystoplasty in the era of onabotulinumtoxinA. Curr Urol Rep 2016;17:27. PMID: 26902621



16. Gao Y, Liao L. Intravesical injection of botulinum toxin A for treatment of interstitial cystitis/bladder pain syndrome: 10 years of experience at a single center in China. Int Urogynecol J 2015;26:1021-6. PMID: 25690160



17. Lin CC, Kuo HC. Video-urodynamic characteristics and predictors of switching from botulinum neurotoxin a injection to augmentation enterocystoplasty in spinal cord injury patients. Toxins (Basel) 2022;14:47. PMID: 35051024



18. Hoebeke P, De Caestecker K, Vande Walle J, Dehoorne J, Raes A, Verleyen P, et al. The effect of botulinum-A toxin in incontinent children with therapy resistant overactive detrusor. J Urol 2006;176:328-31. PMID: 16753434


19. Schulte-Baukloh H, Michael T, Stürzebecher B, Knispel HH. Botulinum-A toxin detrusor injection as a novel approach in the treatment of bladder spasticity in children with neurogenic bladder. Eur Urol 2003;44:139-43. PMID: 12814690


20. Chen G, Liao L. Injections of botulinum toxin A into the detrusor to treat neurogenic detrusor overactivity secondary to spinal cord injury. Int Urol Nephrol 2011;43:655-62. PMID: 21110094



21. Reitz A, Stohrer M, Kramer G, Del Popolo G, Chartier-Kastler E, Pannek J, et al. European experience of 200 cases treated with botulinum-A toxin injections into the detrusor muscle for urinary incontinence due to neurogenic detrusor overactivity. Eur Urol 2004;45:510-5. PMID: 15041117


22. Hassouna T, Gleason JM, Lorenzo AJ. Botulinum toxin A’s expanding role in the management of pediatric lower urinary tract dysfunction. Curr Urol Rep 2014;15:426. PMID: 24903352



23. Kajbafzadeh AM, Ahmadi H, Montaser-Kouhsari L, Sharifi-Rad L, Nejat F, Bazargan-Hejazi S. Intravesical electromotive botulinum toxin type A administration--part II: clinical application. Urology 2011;77:439-45. PMID: 20800886


24. Marte A. Onabotulinumtoxin A for treating overactive/poor compliant bladders in children and adolescents with neurogenic bladder secondary to myelomeningocele. Toxins (Basel) 2012;5:16-24. PMID: 23274271



25. Padmanabhan P, Scarpero HM, Milam DF, Dmochowski RR, Penson DF. Five-year cost analysis of intra-detrusor injection of botulinum toxin type A and augmentation cystoplasty for refractory neurogenic detrusor overactivity. World J Urol 2010;29:51-7. PMID: 21110030



26. Anquetil C, Abdelhamid S, Gelis A, Fattal C. Botulinum toxin therapy for neurogenic detrusor hyperactivity versus augmentation enterocystoplasty: impact on the quality of life of patients with SCI. Spinal Cord 2016;54:1031-5. PMID: 27112841



Fig. 1
The VUDS results at baseline and final follow-up. (A, B) The VUDS showed that MBC (from 72 mL to 230 mL) and BC (from 5.54 mL/cm H2O to 11.25 mL/cmH2O) increased while MDP (from 30 cm H2O to 20 cm H2OH2O) decreased after BTX-A injection treatments, and an improvement in left VUR was not observed. (C, D) The VUDS showed that MBC (from 44 mL to 500 mL) and BC (from 6.08 mL/cm H2O to 50.00 mL/cm H2O) increased while MDP (from 21 cm H2O to 10 cm H2O) decreased after AUEC, and no VUR was detected. VUDS, video-urodynamic study; MBC, maximum bladder volume; BC, bladder compliance; MDP, maximum detrusor pressure; Scr, serum creatinine; BTX-A, botulinum toxin-A; AUEC, augmentation uretero-enterocystoplasty; VUR, vesicoureteral reflux.
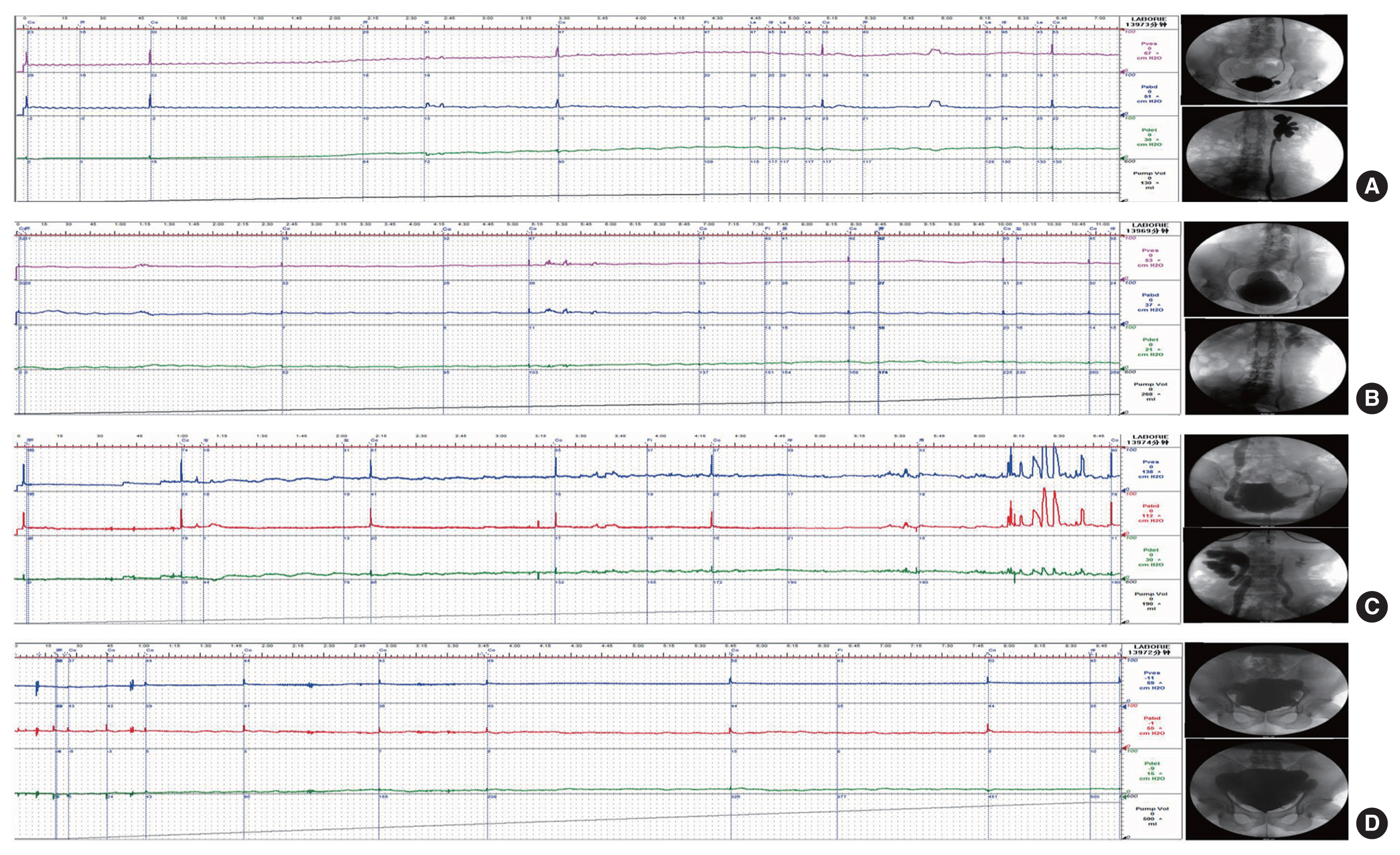
Fig. 2
Improvements in urodynamic parameters for the BTX-A injection group (A) and AUEC group (B) and an adjusted smooth curve fitting analysis of the urodynamic parameters (C–E) during follow-up in both groups. BTX-A, botulinum toxin-A; AUEC, augmentation uretero-enterocystoplasty. The general additive mixed models were adjusted for age, sex, disease duration, vesicoureteral reflux, detrusor overactivity, and incontinence. *P<0.05 when compared with pretherapy data.
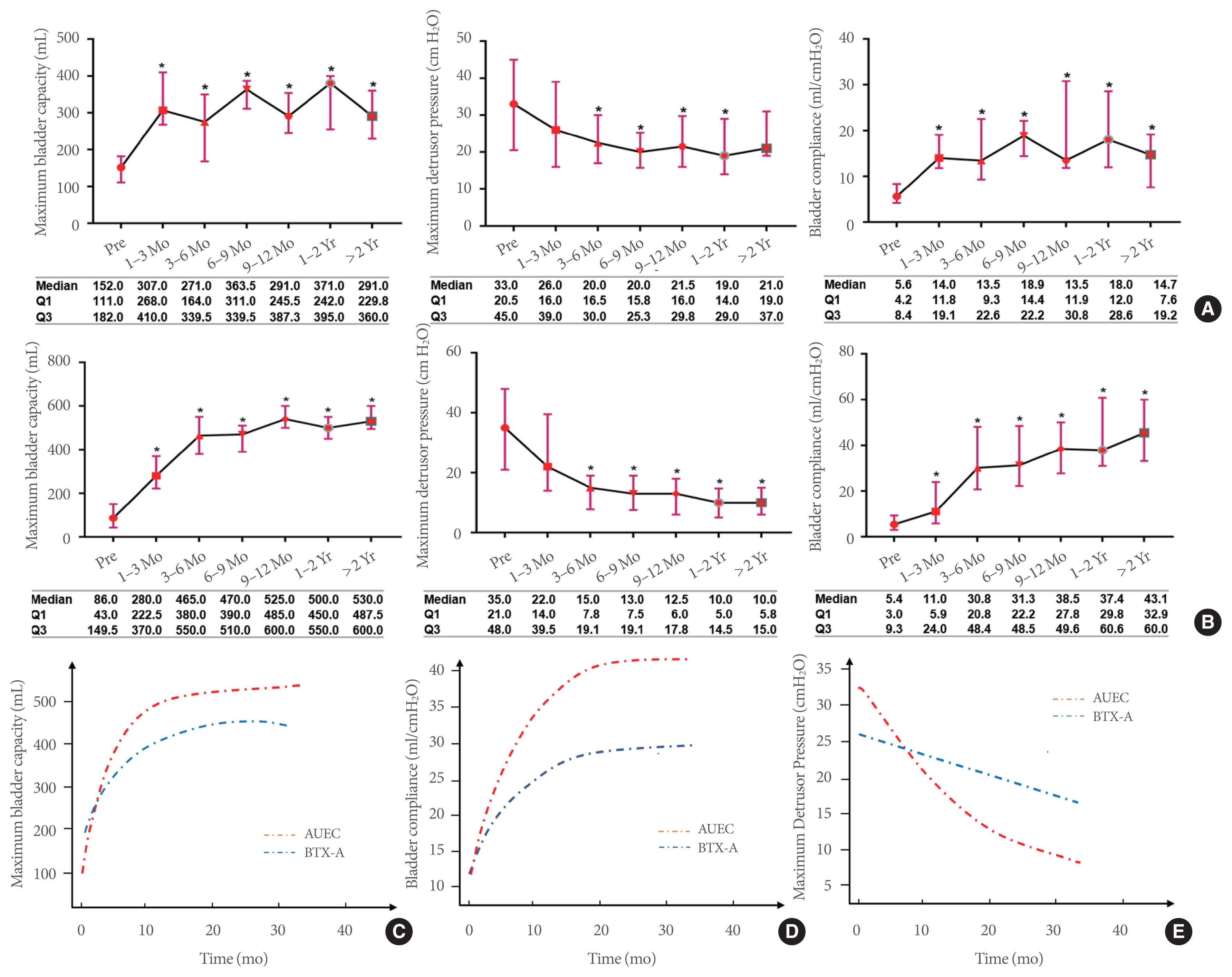
Fig. 3
A Sankey diagram of the grade distribution of VUR and UUTD in the BTX-A injection (A, B) and AUEC (C, D) groups. (A) Changes in the grade distribution of reflux in all ureters at baseline and last follow-up, (B) changes in grade distribution for the MRU-UUTD at baseline and last follow-up, (C) changes in the grade distribution of VUR at baseline and last follow-up, and (D) changes in the grade distribution of UUTD at baseline and last follow-up. VUR, vesicoureteral reflux; UUTD, upper urinary tract dilatation; BTX-A, botulinum toxin-A; AUEC, augmentation uretero-enterocystoplasty; MRU, magnetic resonance urography
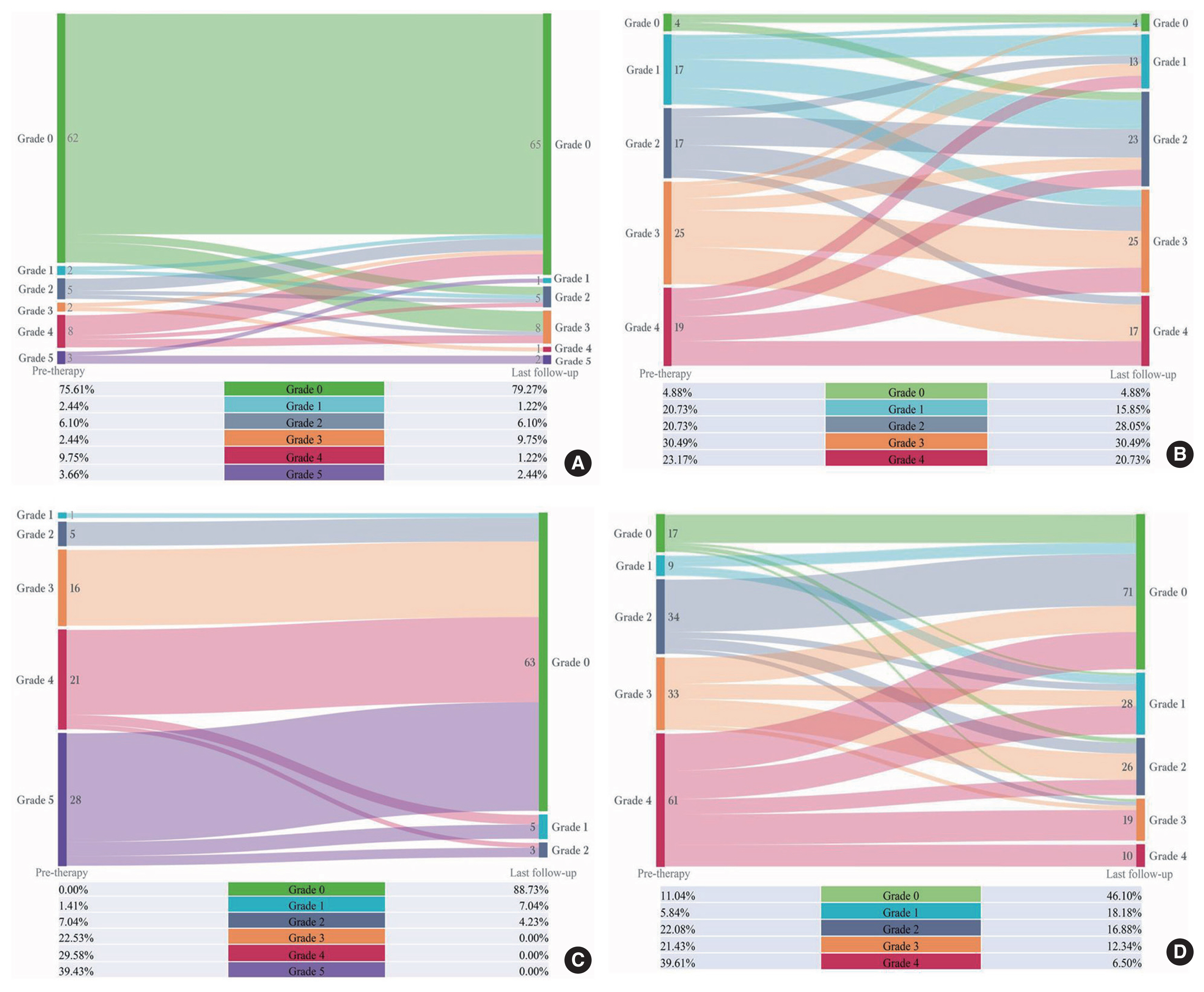
Fig. 4
Changes in Scr and eGFR levels in the BTX-A injection and AUEC groups. (A, D) The changes in Scr and eGFR levels over time in patients with BTX-A injection therapy. (B, E) The changes in Scr and eGFR levels over time in patients who underwent AUEC. (C, F) The adjusted smooth curve fitting analysis of the Scr and eGFR levels during follow-up in the BTX-A and AUEC groups. The general additive mixed model was adjusted for age, gender, disease duration, VUR, DO, and incontinence. Scr, serum creatinine; eGFR, estimated glomerular filtration rate; BTX-A, botulinum toxin-A; AUEC, augmentation uretero-enterocystoplasty; VUR, vesicoureteral reflux; DO, detrusor overactivity.
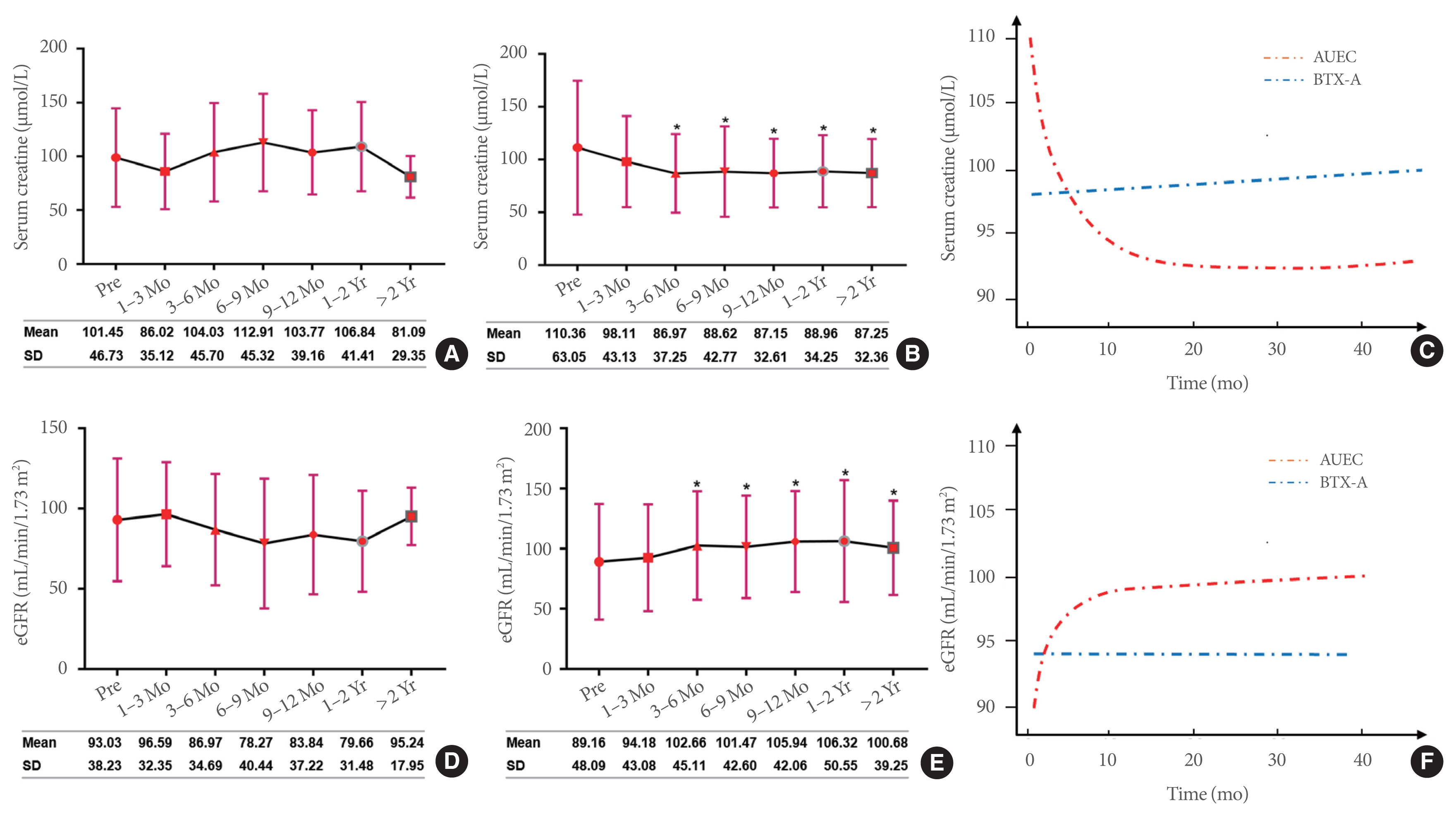
Fig. 5
Comparison of the pre- and posttherapeutic MRU for UUTD in the BTX-A injection and AUEC patient groups. (A, B) MRU-UUTD did not show a significant improvement following BTX-A injection; (C, D) MRU-UUTD showed a significant improvement following AUEC. MRU, magnetic resonance urography; UUTD, upper urinary tract dilatation; BTX-A, botulinum toxin-A; AUEC, augmentation uretero-enterocystoplasty.

Table 1
The demographic and clinical characteristics of patients receiving BTX-A injection and AUEC
| Characteristic | BTX-A (N=41) | AUEC (N=80) | P-value |
|---|---|---|---|
| Age (yr) | 33.37±14.29 | 28.77±13.77 | 0.09 |
|
|
|||
| Follow-up duration (mo) | 17.00 (8.30–21.00) | 24.00 (6.50–26.00) | 0.21 |
|
|
|||
| Disease duration (yr) | 6.00 (3.50–15.00) | 14.50 (3.50–22.00) | 0.06 |
|
|
|||
| Scr (μmoI/L) | 101.45±46.73 | 110.36±63.05 | 0.43 |
|
|
|||
| Left renal pelvis separation | 1.90 (1.40–2.60) | 2.00 (1.28–2.85) | 0.81 |
|
|
|||
| Right renal pelvis separation | 1.90 (1.10–2.00) | 1.60 (080–2.32) | 0.61 |
|
|
|||
| MBC (mL) | 152.00 (112.00–176.00) | 86.00 (43.50–148.00) | <0.01 |
|
|
|||
| MDP (cm H2O) | 33.00 (21.00–44.00) | 35.00 (21.50–48.00) | 0.69 |
|
|
|||
| BC (mL/cm H2O) | 5.62 (4.27–8.00) | 5.43 (3.04–9.27) | 0.38 |
|
|
|||
| Sex | 0.72 | ||
| Male | 29 (70.73) | 54 (67.50) | |
| Female | 12 (29.27) | 26 (32.50) | |
|
|
|||
| Incontinence | 0.81 | ||
| No | 24 (58.54) | 45 (56.25) | |
| Yes | 17 (41.46) | 35 (43.75) | |
|
|
|||
| DO | 0.56 | ||
| No | 26 (63.41) | 55 (68.75) | |
| Yes | 15 (36.59) | 25 (31.25) | |
|
|
|||
| VURa) | <0.01 | ||
| No | 62 (75.61) | 83 (53.90) | |
| Yes | 20 (24.39) | 71 (46.10) | |
|
|
|||
| Etiology | - | ||
| Spinal cord injury | 22 (53.66) | 16 (20.00) | |
| Spinal bifida | 4 (9.76) | 14 (17.50) | |
| Tethered cord syndrome | 5 (12.20) | 13 (16.25) | |
| Myelomeningocele | 2 (4.88) | 19 (23.75) | |
| Others | 1 (2.44) | 5 (6.25) | |
| Unclear | 7 (17.07) | 13 (16.25) | |
Table 2
Unadjusted and adjusted rates of change in MBC, MDP, BC, and Scr among BTX-A injection and AUEC groups within 15 months after GAMMs analysis
| Characteristic | MBC | MDPa) | BC | Scr | ||||
|---|---|---|---|---|---|---|---|---|
|
|
|
|
|
|||||
| β | P-value | β | P-value | β | P-value | β | P-value | |
| Unadjusted | ||||||||
| BTX-A | 14.81 | <0.01 | −0.83 | <0.01 | 1.11 | <0.01 | −0.15 | 0.60 |
| AUEC | 32.15 | <0.01 | −2.01 | <0.01 | 3.19 | <0.01 | −1.42 | <0.01 |
| Difference | 17.51 | <0.01 | −1.17 | <0.01 | 2.05 | <0.01 | −1.28 | <0.01 |
|
|
||||||||
| Adjusted | ||||||||
| BTX-A | 13.62 | <0.01 | −0.68 | <0.01 | 1.09 | <0.01 | 0.02 | 0.96 |
| AUEC | 22.67 | <0.01 | −1.45 | <0.01 | 2.82 | <0.01 | −1.09 | <0.01 |
| Difference | 13.20 | <0.01 | −1.09 | <0.01 | 1.99 | <0.01 | −1.36 | <0.01 |
The model was adjusted for age, sex, disease duration, vesicoureteral reflux, detrusor overactive, and incontinence.





