INTRODUCTION
The standardization of urodynamic evaluations serves both as a boon and a curse to current day clinical neurourology. In many cases, the evaluation of a clinical case of dysfunction in urination progresses no further than identifying the abnormal spinal reflexes gleaned from bladder and abdominal pressure sensors, and the accompanying perineal myogenic response. Seldom does a clinical study for novel pharmaceuticals progress beyond the bladder mucosal receptor, and the entirety of discussion surrounding basic clinical pharmacology in urology often ends with whether the substance crosses the blood brain barrier.
Despite these drawbacks, neurourology has progressed slowly and steadily since the days when the voiding nuclei were first identified and nominated, if not clinically, then at least at the bench. Recent developments have taken advantage of optogenetics, the marriage of genetic manipulation to neurology which allowed selective control over distinct nuclei, to build upon the developments begun more than a hundred years ago in the early ages of neuroanatomy.
This review aims to reiterate classical concepts of voiding in the language of today’s new developments in the science of neurourology.
LOWER MOTOR NEURONS
1. Autonomic Neurons and the Internal Sphincter
The principal components of voiding and storage comprise of voluntary and autonomous components. The autonomous components are commonly described by their gross anatomical designations which convey the organ-based modular understanding which has already been understood in depth for nearly 200 year [1]. The understanding, and also the naming, of the muscular portion of the bladder wall, its layers and the general concept of its function has been generally in agreement since the times of Galen, to Vesalius and to the present day.
The internal sphincter, however, has faced much contention through its history, as more classical texts have described a clear sphincter-like arrangement [2]. Of note, however, Griffiths observes that, while there is no definite sphincter-like arrangement in the bladder neck, compared to other animals, the functional internal sphincter does not terminate at the bladder, but is of a contiguous system which extends down to the urethra, with humans extending to about half an inch, whereas in cats the length may reach up to 2 inches. Unfortunately, these teachings are not well appreciated, and even modern textbooks often depict the internal sphincter as a notable bulging muscle group at the neck of the bladder [3]. As such, modern anatomists are constantly required to ‘rediscover’ the functional internal sphincter in humans and animals [4,5].
In this context, the distinction to diverge from organ-based anatomy and embrace a more functional understanding seems prudent. Therefore, in this paper, which focuses more on the neurological control of these organs, it is suffice to consider the division among only the voiding and storage mechanisms controlled by autonomic functions on one part, and the voluntary control exerted on the external urethral sphincter (EUS).
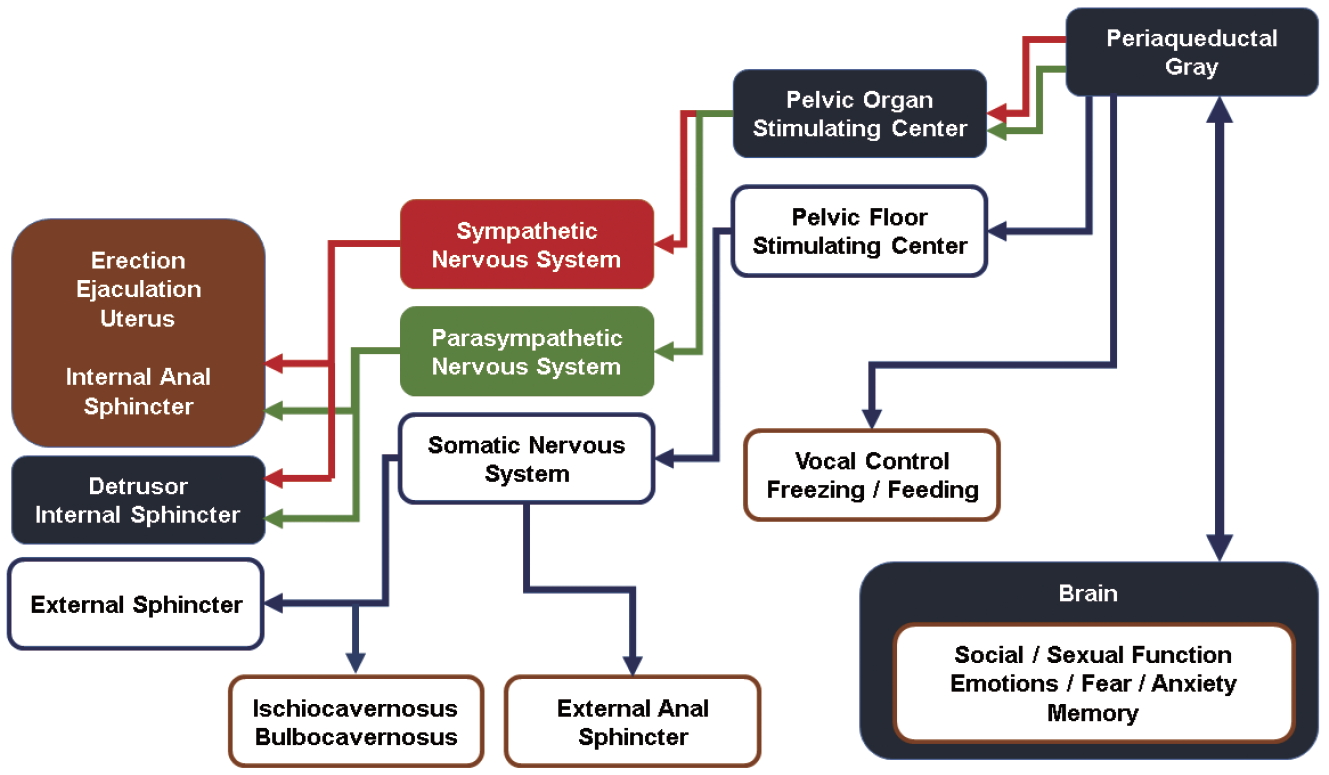
The autonomic control of voiding and storage is maintained by the sympathetic and parasympathetic nerves. The sympathetic function of voiding is maintained by neurons originating in the lateral horn of thoracolumbar cord segments T11 through L2 of the overall thoracolumbar outflow, which encompasses T1 to L2 [6]. The parasympathetic outflow neurons are located in the sacral (S2-4) segments of the overall craniosacral parasympathetic outflow (which includes the cranial nerves 3, 7, 9, and 10). By principle, the sympathetic ganglia connect the preganglionic and post ganglionic neurons at the paraspinal ganglia, whereas the parasympathetic ganglia are terminal or intramural ganglia located in proximity to the bladder, in this case the inferior hypogastric plexus (IHP) [7,8].
It is often taken as a binary division that the sympathetic outflow passes through the hypogastric nerves, whereas the parasympathetic outflow is associated with the pelvic splanchnic nerves. In actuality, post ganglionic divergences of neurons range from 4 to 20, resulting in several small microscopic fibers that could not definitely be determined its origin without advanced methods [9]. Recently, Alsaid et al. [10] showed coexistence of both parasympathetic and sympathetic fibers for both courses. Li et al. [11] showed the scant paucity and the delicate nature of these neurons scattered within the IHP, consisting about 4% of the cross section. Modern investigations of the IHP underscores the significant lack of tools available to classical anatomists to understand functional neurourology.
As such, the general understanding is that the end organ detrusor muscles, contiguous from the bladder to the urethra, are autonomically innervated in function by parasympathetic and sympathetic outflows.
2. Onufrowicz’s Nucleus and the EUS
In contrast to the autonomic voiding/storage apparatus and its innervation, the somatic control of voiding and the EUS remain distinct. There has been no major misunderstanding concerning the presence of an EUS. While the muscular structure has always been apparent, the discovery of distinct nerve cells by the Polish American Bronislaw Onufrowicz (Onuf for short) had been fortuitous [12]. Onuf had noted the distinct morphology of the nuclei and had further associated its function to the bulbocavernosus and ischiocavernosus muscles, concluding its primary function in penile erection. It was only until latter studies that the range of its functional control expanded to the anal and externa urethral sphincters [13-16]. The neurons from Onuf’s nucleus maintains continence of micturition and defecation through the perineal nerves [17].
Modern understanding of the EUS via Onuf’s nucleus involves a complex interplay between the autonomic and somatic nerves. Even though these neurons participate in the process of signal transmission through similar basic mechanisms as other neurons—via neurotransmitters that are secreted by the presynaptic neuron and bind with postsynaptic receptors—their axons contain fewer dense core vesicles and secretory granules than are found in autonomic nerves [18]. The motor neurons of these nuclei are rich in serotonin (5-HT) and norepinephrine (NE) receptors and are activated by L-glutamate, a neurotransmitter that is the anionic form of glutamic acid. If activated by 5-HT and NE, the guarding reflex, which suppresses urination, occurs and prevents urination when abdominal pressure suddenly increases.
Regarding incontinence, a 3-layer muscle is involved in urine flow and continence that consists of an inner band of longitudinal smooth muscle, a middle band of circular smooth muscle, and a striated muscle known as the rhabdosphincter. This muscle is dominated by sympathetic neurons, parasympathetic neurons, and the somatic branch of the peripheral nervous system. First, sympathetic neurons are distributed in the longitudinal and circular smooth muscle layers, running along the hypogastric nerve and synapsing with sympathetic preganglionic neurons located in the upper lumbar spinal cord [17]. Parasympathetic neurons arise from parasympathetic preganglionic neurons located in the sacral spinal cord and are also distributed in the longitudinal and circular smooth muscle layers. Somatic nerves originate from motor neurons located in the ventral or anterior horn of the sacral spinal cord, i.e., Onuf’s nucleus. The pudendal nerve also extends from Onuf’s nucleus and directly controls the rhabdosphincter. The sympathetic storage reflex, which is also called the pelvic-to-hypogastric reflex, starts when NE is secreted in response to bladder stretching [19]. The somatic storage reflex, which known as the pelvic-topudendal or guarding reflex, occurs in situations such as laughter, sneezing, and coughing that induce a rise in vesical pressure due to increased abdominal pressure or detrusor pressure. L-Glutamate, which is the primary excitatory transmitter in the reflex arc, triggers action potentials by stimulating N-methylD-aspartate, α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid, and transmembrane ionotropic receptors, and these action potentials stimulate the secretion of acetylcholine, thereby causing contraction of rhabdosphincter fibers [20]. It is thought that stress urinary incontinence occurs if this guarding reflex does not function properly.
The coordination between the somatic control of the EUS and the autonomic control of the detrusor muscles is an interesting phenomenon. While the actual act of voiding is an entirely autonomic execution of a set of responses, the initiation itself is entirely by somatic voluntary command. Similar associations exist in several other voluntary to autonomic responses, including swallowing, where the inferior constrictor (IC) muscle and the cricopharyngeal (CP) muscles interplay in a transfer of hands similar to voiding reflexes [21,22]. Different subtypes of amyotropic lateral sclerosis (ALS) can be seen to mirror these changes, as bulbar ALS involves speech and dysphagia, whereas spinal ALS is also known to involve Onuf’s nucleus, thereby eliciting voiding disorders [23]. The connection from somatic to autonomic is most apparent during urodynamic studies, where the silencing of pelvic electromyographic activity before voiding is interrupted with intermittent bursts of EUS activity. Similar activity can also be seen between IC and CP during swallowing [24]. Where the similarity ends is also interesting, as the act of swallowing begins actively in amnio, while voiding is acquired in the early years of infancy. Hence, several studies have demonstrated a period of learning during which the electromyography signal gradually become suppressed during the reflexive voiding phase, only to reappear following spinal injury, as noted in cases of detrusor sphincter dyssynergia [6,25,26]. The plasticity of interneurons develops towards control from the upper motor neurons (UMNs), primarily from Barrington’s nucleus.
UPPER MOTOR NEURONS
1. Barrington’s Nucleus, the M-Region, and the Pelvic Organ Stimulating Center
Barrington’s nucleus is located medial to the locus coeruleus in the pons. Its principal function was identified during discovery by F. J. F. Barrington, a urologist at University College Hospital, London, through a series of experiments published during the 1920s; borrowing the then novel stereotaxic surgical equipment from its pioneering developers, physiologist Robert Clarke and neurosurgeon Victor Horseley, Barrington performed needle destruction studies on cats whilst documenting its corresponding functional voiding changes [27].
Although, Barrington had discovered his eponymous nucleus with the intent to uncover the voiding center, neuroscience has since identified a variety of different populations within Barrington’s nucleus, with some interesting topographic distinctions as well. The principal neurons in regards to micturition have been identified to be corticotropin-releasing factor-positive, which project to the spinal parasympathetic nucleus innervating the bladder [28]. Holstege et al. [29] distinguished Barrington’s nucleus as the M-region, in contrast to a more lateral field (the L-region) which controlled Onuf’s nucleus, which would then, in turn, activate the EUS and other pelvic floor muscles, not only limiting to micturition and storage, but also to defecation and sexual activity.
The most recent terminology presents Barrington’s nucleus as the pelvic organ stimulating center (POSC), as neurons in this group not only project to parasympathetic neurons eliciting micturition, but also to parasympathetic neurons to the uterus, the distal colon and rectum. In contrast, the L-region, caudal and ventrolateral (VL) to the POSC is termed the pelvic floor stimulating center (PFSC), its stimulation activating the pelvic floor in general, via Onuf’s nucleus, including the EUS and, returning to Onuf’s original discovery, the bulbocavernosus and ischiocavernosus muscles [29-33]. Since Holstege [31] seems to have abandoned the terms M- and L-region for POSC and PFSC, respectively, the most modern nomenclature would be for the latter (Fig. 1).
2. The Periaqueductal Gray
The earliest studies on the periaqueductal gray (PAG) followed the seminal works of Barrington, by introducing lesions in cats [34]. Earlier studies focused both on micturition and the apparent psychological aspects that destruction of associated structures induced. The overall function of the PAG in total is too broad to cover in this review. However, the study of the PAG has been partitioned to 4 general anatomical locales that seem to have distinct functional properties: dorsomedial, dorsolateral (DL), lateral, and VL, which have distinct connections with the forebrain, brainstem, and nociceptive neurons of lamina I of the spinal cord and trigeminal nucleus. Concerning micturition, the DL and VL columns have been highlighted.
Whereas the pons has presented relatively centralized pathways for both voiding and active retention in POSC and PFSC, respectively, research into the PAG micturition control has shown an increasing tendency towards interconnection with complex functions. While the DL column projects modulatory gamma-aminobutyric acid signals, and the VL column projects primary glutamate signals to the POSC, each column also integrates various cranial pathways such as emotional, vocalization and motor functions [33,35-38]. The PAG shows a wide range of integration with various subsystems, including freezing vs. feeding in association with the cerebellum, sex and pain response, and vocal control [39-42].
PAG is distinct from the POSC in that sensory information on bladder filling passes through Aδ fibers from detrusor muscles coursing through the dorsal horn and up the spinal cord are integrated here. In rats, this connection allows direct command to the POSC to initiate micturition [43]. However, in humans, and cats and dogs and other primates, the PAG does not decide micturition based on bladder filling information alone, and are subject to many brain structures [31,44]. Interesting, though, rats have also shown some degree of social modulation of stress retention [45,46]. As such, the difference between how the PAG integrates both peripheral signals with higher complex functions to direct voiding in the POSC and PFSC in humans and rats would be an interesting subject in understanding how our social and emotional controls of voiding develop. As such, Holstege [47] has termed this function of the PAG as the Emotional Motor System.
3. Central Pathways
The complex, but incomplete, understanding of the brain in control of micturition implicates various structures involved in the decision on when and where to void [48,49]. There is an understandable lack of concrete evidence, as presented so far in studies up to the mid brain. Most recent studies focus on functional magnetic resonance images (fMRI) limited to only a handful of subjects [50,51]. While the sensation of bladder distention and urgency has been associated strongly with the thalamus in rats, human studies show involvement of a plethora of other structures [52-55].
The hypothalamic preoptic area projects to the POSC, conferring a “safe signal” that it is permissible to void [36,56,57]. During voluntary urine withholding, blood flow to the cingulate gyrus significantly decreases, while the prefrontal cortex is active when micturition is taking place during involuntary withholding [58-61].
However, whereas the specific control of voiding and retaining urine generally ends with the motor control of the PAG, and then to the POSC and PFSC, higher functions are now dictated to various conscious controls spread out through the brain. The integrative and centralized tendency seen so far progressing, from the periphery to the pons and midbrain, now expands to receive input from multiple other functional components, each integrating in itself complex interactions. Controlled experiments are difficult, as available experimental animal cannot be relied on to share similar structures and connections. Further progress, thus, required an entirely new toolset of investigation.
4. Directions of Neurourological Research
In the aging process or in disease states accompanied by neurological disorders, the cerebral control of suppression is weakened due to unnecessary activation of the micturition center. Nocturnal enuresis and accompanying lower urinary tract symptoms are sometimes thought to be caused by spinal reflexes that are not suppressed and controlled in the cerebrum. Therefore, if external local effects on inappropriate activation of the micturition center in the brain can be modified, these patterns of inappropriate activation can in turn be controlled, and may even be the target of treatment [62]. If confirmed, this is a promising possibility for our understanding of diseases accompanied by dysuria, such as aging and degenerative brain diseases, and it may facilitate the development of drugs to treat those diseases.
Currently, urodynamic studies are used for the definitive establishment of the phenotypic cause of disorders when diagnosing and treating patients with lower urinary tract dysfunction, including neurogenic dysfunction. Urodynamic studies enable a determination of whether the phenotype is myogenic, urotheliogenic, or neurogenic, and if it is neurogenic, they can shed light on the location of damage. Subsequently, any controllable pathophysiological factors related to the patient’s condition are found and addressed as part of the treatment plan. However, this treatment strategy is only applicable to peripheral treatment targets. Therefore, an integrated research framework exploring the factors that hinder central inhibition in the micturition center, as discussed above, may lead to the identification of various methods to control factors that promote dysfunction. In other words, it is necessary to find tools for exploring the neurogenic phenotype.
The central focus of investigative tools reviewed thus far within this article shows how the discipline of neurourology is dependent upon understanding the specific compositional properties, and integrating this knowledge into the overall phenotypic expression of micturition and storage. The study of specific investigations has progressed from anatomical dissection to stereotactic lesions to rabies virus-mediated tracing, with the most advanced iteration presenting as the new science of optogenetics.
Optogenetics refers to the integration of optics and genetics to achieve gain or loss-of-function of well-defined events within specific cells of living tissue [63]. Discovery of bacterial opsin genes and rhodopsin proteins are distinct from vertebrate counterparts in that they do not couple to secondary messengers to influence ion channels, but directly transduce photons into electrical currents [64]. Though the initial discovery of rhodopsin-like proteins was expressed in bacteria had been made in the early 1970s, the technology to genetically clone expression to select neurons and then to be able to deliver select frequencies of light required significant developmental challenges [65-67]. This new technology has gained rapid traction during the past decade. The recent trends of research delve into both the UMN and the LMN, investigating the spinal cord, the micturition centers of the pons and midbrain, as well as the pelvic neural ganglia [68-70] (Figs. 2, 3).
From an alternative perspective, physiological animal models which interpreted controlled experiments of dissected strips or anaesthetized animals can now be understood with fMRI images [71]. Artificial intelligence (AI) can be utilized from providing sharper images to conventional or novel techniques, control and perform complex optogenetic signal modulations, or simulate in silico models of control pathways. Modern development of AI has always reflected some aspect of neurology, whether it was from a ground-up development for the neural network-based perceptrons evolving into complex deep networks, or Bayesian-based symbolic AI developing from a relatively top-down position. However, the utility of AI in neuroscience is in itself too vast a topic to be covered in this review.
Yet, these methods are yet in their infancy, and are still limited when dealing with complex behavioral activity when it comes to humans. The basic mechanisms underlying these relationships have not been identified, meaning that specific methods to control such changes have not been developed. Observing and testing the roles of specific neurotransmitters and neuropeptides in controlling urination in the brain by activating and suppressing synaptic connections will make vital contributions to research into basic mechanisms in the field of neurourology [72]. Future investigations also point to an integrative approach.
CONCLUSIONS
The control of micturition is not isolated. The LMNs are interconnected with all pelvic functions from bowel movement to sexual stimuli, while the UMNs are deeply associated with emotions, social behavior, and pain. The association, not only with neurology and urology, but with speech rehabilitation, psychiatry, proctology, and sexual medicine opens up a complex avenue for investigation.