Irreversible Bladder Remodeling Induced by Fibrosis
Article information
Abstract
Underactive bladder and impaired bladder compliance are irreversible problems associated with bladder fibrosis. Remodeling of the extracellular matrix is regarded as an important mechanism associated with bladder fibrosis. However, various risk factors and conditions contribute to the functional impairment of the bladder associated with fibrosis, and there is limited knowledge about bladder fibrosis-associated problems in the field of neurourology. Further studies are thus necessary to elucidate the underlying mechanism of bladder fibrosis and to identify effective treatment.
INTRODUCTION
Bladder trabeculation refers to morphological changes of the bladder detrusor, including smooth muscle hypertrophy and increased collagen deposition in the detrusor extracellular matrix (ECM), resulting in fibrosis of the bladder [1,2]. These morphological changes of the bladder can be observed in patients with various voiding problems, such as neurogenic bladder and bladder outlet obstruction (BOO) [3-6]. Lower urinary tract symptoms (LUTS) in the men with benign prostatic hyperplasia (BPH) are associated with BOO, and the increased intravesical pressure that occurs in men with BOO induces hypertrophy of the bladder detrusor to overcome BOO. If BOO is not relieved, irreversible morphological changes of the bladder, such as increased collagen accumulation and fibrosis of the bladder, occur. Fibrosis of the bladder causes the loss of normal detrusor contractility; and therefore, affected patients cannot urinate by themselves [6]. In addition to BOO associated with BPH, many other conditions are associated with fibrotic changes of the bladder, such as dementia, stroke, cerebral hemorrhage, spinal cord injury, diabetes mellitus, and aging [7].
Common LUTS associated with bladder fibrosis are a weak urinary stream, intermittency, increased residual urine sensation, and abdominal straining during urination because the fibrotic bladder loses normal contractility for expelling urine from the bladder. Medical therapies using parasympathomimetics and alpha blockers have been attempted to help urination; however, inconsistent results have been reported regarding the effects of these treatments, and there is no clear evidence that they improve bladder contractility [8-11]. Some patients showed improvement of LUTS and the ability to urinate by themselves, but most patients continue to need indwelling urethral and suprapubic catheters to expel urine from their bladder due to an irreversible loss of bladder contractility [12-15].
At present, no effective treatment methods are available to prevent bladder fibrosis and to recover the impaired bladder contractility associated with bladder fibrosis. Thus, this review deals with the pathophysiology of bladder fibrosis and upcoming treatment based on a literature review.
LUTS AND DECREASED CONTRACTILITY AND COMPLIANCE OF THE BLADDER DETRUSOR ASSOCIATED WITH BLADDER FIBROSIS
Underactive bladder (UAB) is a LUTS complex characterized by incomplete bladder emptying with a decreased urinary flow rate and increased postvoid residual urine volume [15]. UAB is associated with detrusor underactivity (DU) or acontractile detrusor observed in a urodynamic study. Morphological changes such as bladder fibrosis induced by increased collagen deposition in the detrusor ECM can induce DU and impairment of bladder contractility. According to a study reporting urodynamic results in UAB patients, detrusor hyperreflexia with impaired detrusor contractility (DHIC) was observed, as well as DU or acontractile detrusor [16]. Patients with DHIC experienced storage symptoms such as incontinence or urgency with accompanying symptoms induced by incomplete emptying [17]. These findings suggest that contractile changes associated with bladder fibrosis constitute a complicated process that results in impaired bladder contractility with or without detrusor overactivity. Furthermore, the process of a coexistent voiding problem that is not associated with bladder fibrosis (e.g., stress urinary incontinence) can be a factor that induces incontinence [18-20]. Moreover, Uren et al. [21] reported that patients diagnosed with DU using a urodynamic study showed nocturia, frequent daytime urination, urgency, and incontinence, as well as a weak urinary stream, hesitancy, and abdominal straining during urination. However, Uren et al. [21] did not investigate the etiology of DU, making it impossible to know whether the patients with storage LUTS had other voiding problems unrelated to DU.
Decreased bladder compliance is a change associated with bladder fibrosis. Bladder compliance can be conceptualized as the relationship between a change in bladder volume and a change in detrusor pressure. In general, bladder compliance is expressed as an increase in bladder volume per increment of intravesical pressure [22]. Thus, bladder compliance reflects the flexibility of the bladder. Fibrotic changes of the bladder reduce its flexibility and induce stiffness of the bladder and low bladder compliance. As a result, the intravesical pressure of a stiff bladder with low compliance is increased, and prolonged high intravesical pressure adversely affects renal function [23-26]. Therefore, it is important to improve bladder compliance and reduce the intravesical pressure of the bladder to prevent impairment of renal function. Patients with impaired bladder compliance show urge urinary incontinence [27].
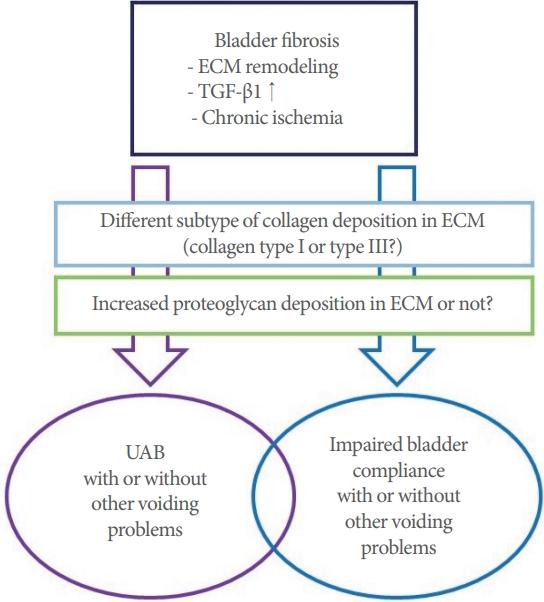
Both UAB and impaired bladder compliance are associated with bladder fibrosis. However, it is unclear about why the consequences of bladder fibrosis sometimes appear as UAB or impaired bladder compliance. Previous studies have suggested that there may be a difference in the subtype of collagen deposition between UAB and impaired bladder compliance, and that increased proteoglycan deposition in the ECM may also be related factor (Fig. 1) [28-31].
UNDERLYING MECHANISM ASSOCIATED WITH BLADDER FIBROSIS
Remodeling of the ECM and increased levels of transforming growth factor-β1 (TGF-β1) are regarded as the mechanism underlying bladder fibrosis [32]. The ECM of the bladder consists of collagen, elastin, fibronectins, and proteoglycans. Collagen is the major fibrous protein of the ECM and plays a role in providing tensile strength to the bladder. Collagen types I and III are the most important subtypes of collagen in the ECM [33- 35]. Unlike collagen, elastin induces recoil of the ECM after stretching during urination. Conditions such as BOO and neurogenic bladder associated with mechanical or chemical stresses induce fibroblastic and inflammatory responses in the ECM. These responses dysregulate fibroblast secretion of matrix metalloproteinases (MMPs) and tissue inhibitors of MMPs (TIMPs) and increase ECM deposition. Prolonged ECM deposition increases the production of TIMPs which are associated with fibrosis and fibrotic changes of the bladder occur [36-38]. Moreover, increased levels of TGF-β1 induce fibrosis by stimulating collagen production through binding to serine/threonine kinase receptors on the cell surface, cause the phosphorylation of intracellular Smad2/3 transcription factors [39,40].
Chronic bladder ischemia may be a factor associated with bladder fibrosis. Previous studies have suggested that cardiovascular and metabolic diseases related to endothelial dysfunction decrease blood flow to the bladder. In turn, chronic ischemia of the bladder was found to increase oxidative stress and inflammatory cytokine levels, which might be associated with bladder fibrosis induced by nerve damage [41].
UPCOMING THERAPEUTIC METHODS TO PREVENT AND TREAT BLADDER FIBROSIS
There is currently no effective treatment for LUTS associated with bladder fibrosis. Conventional medical treatment fails in most patients with voiding problems induced by bladder fibrosis, and these patients require catheterization to expel urine from the bladder. Therefore, studies have aimed to prevent and restore bladder fibrosis using antifibrotic agent such as relaxin. Ikeda et al. [42] showed that human relaxin-2 reversed fibrosis, decreased collagen deposition, and increased bladder compliance and detrusor contractility in patients with radiation-induced bladder fibrosis. A recent study reported that relaxin receptors were present in the dome and trigone of the human bladder and that in vitro relaxin stimulation upregulated MMP- 2 and decreased TGF-β1 [43].
Efforts have been made to apply stem cell and gene therapy to improve UAB associated with bladder fibrosis [20]. Several preclinical studies used various types of stem cells and showed improvements in detrusor contractility. A pilot study by Levanovich et al. [44] showed enhanced urination and a reduced need for clean intermittent catheterization in a patient with UAB after an intradetrusor injection of autologous muscle-derived stem cells.
CONCLUSIONS
Bladder fibrosis is an irreversible change of the bladder that is associated with UAB and impaired bladder compliance. Although bladder fibrosis is a serious problem, it has been difficult to elucidate its exact underlying mechanism and risk factors. Moreover, the progression of voiding problems associated with bladder fibrosis cannot be predicted due to the unknown characteristics of conditions associated with bladder fibrosis. There problematic characteristics are also obstacles to the prevention and treatment of bladder fibrosis. Therefore, further studies are essential to elucidate the underlying mechanism of bladder fibrosis and to identify effective treatments.
Notes
Fund/Grant Support
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2018R1D1A3B07048492).
Conflict of Interest
SJK and KHK, associate editors of International Neurourology Journal, are the authors of this article. However, they played no role whatsoever in the editorial evaluation of this article or the decision to publish it. Except for that, no potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTION STATEMENT
·Conceptualization: SJK
·Data curation: JK
·Formal analysis: YGN
·Funding acquisition: KHK
·Methodology: JK
·Project administration: KHK
·Visualization: YGN
·Writing-original draft: SJK
·Writing-review & editing: SJK