Effects of High Concentrations of Naftopidil on Dorsal Root-Evoked Excitatory Synaptic Transmissions in Substantia Gelatinosa Neurons In Vitro
Article information
Abstract
Purpose
Naftopidil ((±)-1-[4-(2-methoxyphenyl) piperazinyl]-3-(1-naphthyloxy) propan-2-ol) is prescribed in several Asian countries for lower urinary tract symptoms suggestive of benign prostatic hyperplasia. Previous animal experiments showed that intrathecal injection of naftopidil abolished rhythmic bladder contraction in vivo. Naftopidil facilitated spontaneous inhibitory postsynaptic currents in substantia gelatinosa (SG) neurons in spinal cord slices. These results suggest that naftopidil may suppress the micturition reflex at the spinal cord level. However, the effect of naftopidil on evoked excitatory postsynaptic currents (EPSCs) in SG neurons remains to be elucidated.
Methods
Male Sprague-Dawley rats at 6 to 8 weeks old were used. Whole-cell patch-clamp recordings were made using SG neurons in spinal cord slices isolated from adult rats. Evoked EPSCs were analyzed in Aδ or C fibers. Naftopidil or prazosin, an α1-adrenoceptor blocker, was perfused at 100 μM or 10 μM, respectively.
Results
Bath-applied 100 μM naftopidil significantly decreased the peak amplitudes of Aδ and C fiber-evoked EPSCs to 72.0%±7.1% (n=15) and 70.0%±5.5% (n=20), respectively, in a reversible and reproducible manner. Bath application of 10μM prazosin did not inhibit Aδ or C fiber-evoked EPSCs.
Conclusions
The present study suggests that a high concentration of naftopidil reduces the amplitude of evoked EPSCs via a mechanism that apparently does not involve α1-adrenoceptors. Inhibition of evoked EPSCs may also contribute to suppression of the micturition reflex, together with nociceptive stimulation.
• HIGHLIGHTS
- Whole-cell patch-clamp recordings were made using SG neurons in spinal cord slices from rats.
- Naftopidil significantly decreased the peak amplitudes of Ad and C fiber-evoked EPSCs, but not prazosin.
- Inhibition of evoked EPSCs by naftopidil may contribute to suppression of the micturition reflex.
INTRODUCTION
Lower urinary tract symptoms (LUTS) are rather bothersome in elderly men and profoundly affect their quality of life (QoL) [1]. LUTS are generally defined in terms of 3 sets of symptoms: voiding, storage, and postmicturition [2]. Male LUTS often present as a result of benign prostatic hyperplasia (BPH), which involves increased resistance to urinary flow caused by bladder outlet obstruction, increased contractility of smooth muscle of the prostate and urethra [3], detrusor overactivity with denervation, decreased blood flow in the urinary bladder, and elevation of mediators (e.g., adenosine triphosphate and prostaglandins) [4,5]. Patients who are diagnosed with BPH are widely prescribed α1-adrenoceptor blockers, which can reduce voiding and storage symptoms (e.g., the former: slow stream, intermittent stream, and straining; the latter: daytime frequency, nocturia, and urgency). The mechanisms of action of α1-adrenoceptor blockers involve inhibition of the contractile response of smooth muscle in the prostate by endogenously released noradrenaline. In patients with BPH, α1-adrenoceptor blockers reduce prostate tone and bladder outlet obstruction to improve voiding symptoms [6,7], increase blood flow in the urinary bladder to improve storage symptoms [4], and inhibit the release of ATP from the urothelium [8]. Because α1-adrenoceptor blockers can lengthen micturition intervals, afferent sensory nerves are also considered a site of action of α1-adrenoceptor blockers [9,10].
Naftopidil ((±)-1-[4-(2-methoxyphenyl)piperazinyl]-3-(1-naphthyloxy)propan-2-ol) is an α1D/1A-adrenoceptor blocker that improves LUTS suggestive of BPH. Naftopidil has been found to improve subjective complaints, objective symptoms, and QoL associated with male LUTS [11,12]. Naftopidil dosedependently inhibited phenylephrine-induced increases in prostatic pressure in canines when administered intravenously [13] and improved bladder overactivity and impaired bladder blood flow caused by bladder outlet obstruction in rats when administered orally [4]. In addition, naftopidil transiently abolished rhythmic bladder contraction in a dose-dependent manner in rats when administered intrathecally at lumbosacral levels [14]. In BPH patients, storage symptoms are much more bothersome to their QoL than voiding symptoms [1,15]. Suppression of the enhanced micturition reflex stabilizes micturition and the storage function of the urinary bladder.
Accordingly, we studied the mode of action of naftopidil on afferent nerve activity related to storage symptoms. In an experiment using rat spinal cord slices, 30 to 100 μM naftopidil facilitated miniature inhibitory postsynaptic currents (mIPSCs) in substantia gelatinosa (SG) neurons, as seen with the patchclamp procedure [16]. That study determined the mechanism of action of naftopidil on the micturition reflex and supported a previous study showing that naftopidil mediates gamma-aminobutyric acid (GABA)/glycine receptors at the level of the spinal cord [17]. However, the effect of naftopidil on excitatory postsynaptic currents (EPSCs) in SG neurons remains to be elucidated. The goal of this study was to further characterize the mechanism of action of naftopidil using the patch-clamp technique.
MATERIALS AND METHODS
All experiments were carried out in accordance with the Guiding Principles for Care and Use of Animals in the Field of Physiological Sciences of the Physiological Society of Japan and were approved by the local Animal Experiment Committee of Kumamoto Health Science University and Kyushu University. All possible efforts were made to minimize animal suffering and the number of animals used for the studies.
Preparation of Spinal Cord Splices
Spinal cord slices were prepared from adult male rats, and blind whole-cell patch-clamp recordings from SG neurons were performed as described previously in detail [18-20]. Briefly, Sprague-Dawley rats aged 6–8 weeks were deeply anaesthetized with urethane (1.2 g/kg, intraperitoneally), and a lumbosacral laminectomy was subsequently performed. The lumbosacral segments of the spinal cord (L2–S3) with ventral and dorsal roots were removed and placed in ice-cold Krebs solution equilibrated with 95% O2 and 5% CO2. The Krebs solution contained (in mM): NaCl 117, KCl 3.6, CaCl2 2.5, MgCl2 1.2, NaH2PO4 1.2, NaHCO3 25, and glucose 11 (pH, 7.4). After the spinal cord was isolated, the rats were immediately killed by exsanguination under urethane anesthesia. The pia-arachnoid membrane was removed after cutting all the ventral and dorsal roots, except for the L5, L6, or S1 dorsal root on 1 side, and any of them was used. The spinal cord was mounted on a vibratome, and a 550-μm-thick transverse slice with the attached dorsal root was cut. The slice was moved onto a nylon mesh in the recording chamber in a volume of 0.5 mL of Krebs solution, and was completely submerged and perfused with Krebs solution saturated with 95% O2 and 5% CO2 at 37°C±1°C at a flow rate of 10–15 mL/min. The dorsal root was stimulated using a suction electrode (Fig. 1A).

Various patterns of excitatory postsynaptic currents (EPSCs) elicited in substantia gelatinosa (SG) neurons by stimulating the dorsal root attached to adult rat spinal cord slices. (A) Schematic illustration of patch-clamp recording from SG neurons. (B) Typical trace of monosynaptic Aδ fiber-evoked EPSCs. (C) Typical trace of monosynaptic C fiber-evoked EPSCs. (D) Typical traces of monosynaptic Aδ and C fiber-evoked EPSCs.
Whole-Cell Patch-Clamp Recordings From SG Neurons
The SG was precisely identified with transmitted illumination as a relatively translucent band across the dorsal horn in the transverse slice preparations. Blind whole-cell voltage-clamp recordings were made of SG neurons, as described previously [16,20,21]. The patch pipettes were filled with a solution containing potassium gluconate solution (in mM): K-gluconate 135, KCl 5, CaCl2 0.5, MgCl2 2, EGTA 5, HEPES 5, and ATPMg 5 (pH, 7.2). The patch pipettes had a tip resistance of 6–12 MΩ. Series resistance was assessed according to the response to a hyperpolarizing step of 5 mV. This value was monitored during the recording session, and data were rejected if values changed by >15%. Signals were acquired with a patch clamp amplifier (Axopatch 700A, Molecular Devices, Union City, CA, USA). The data were digitized with an AD/DA converter (Digidata 1321A, Molecular Devices), stored on a personal computer using a data acquisition program (Clampex version 9.0, Molecular Devices), and analyzed using a software package (Clampfit version 9.0, Molecular Devices). Cell recordings were made in voltage-clamp mode at holding potentials of -70 mV to record EPSCs [16,20]. To elicit EPSCs, the dorsal roots were stimulated at a frequency of 0.2 Hz (duration, 100 μsec). The Aδ- or C-afferent-mediated responses evoked by dorsal root stimulation were distinguished based on the conduction velocity of the afferent fibers (C-fiber response: <0.8 m/sec; Aδ-fiber response: 2.0–11 m/sec) and stimulus threshold (C-fiber response: >200 μA; Aδ-fiber response: 20–200 μA), as previously described [19,20]. The conduction velocity was calculated from the latency of synaptic responses and the length of the dorsal root. The evoked responses were considered monosynaptic for the Aδ fiber-evoked EPSCs if the latency remained constant when the root was stimulated at 20 Hz, and for the C fiber-evoked EPSCs if no failure occurred regardless of the constancy of the latency when stimulated at 2 Hz [19,20]. To avoid the influence of evoked release on presynaptic terminals, dorsal root stimuli were not applied during the examination of the effects of drugs on spontaneous or miniature synaptic responses.
Drug Application
Naftopidil (PubChem CID: 4418) (Asahi Kasei Pharma Co., Tokyo, Japan) was dissolved in 1% dimethyl sulfoxide (DMSO) (PubChem CID: 679) (Wako, Osaka, Japan) in Krebs solution. The other drugs were dissolved in Krebs solution and applied by perfusion via a 3-way stopcock without changes in the perfusion rate or temperature. The other drug used in this study was prazosin hydrochloride (PubChem CID: 68546) (Wako).
Statistical Analysis
All data values are shown as the mean±standard error of the mean. The differences were compared using the Student paired and unpaired t-tests. P-values<0.05 were considered to indicate statistical significance, and n refers to the number of neurons measured.
RESULTS
Monosynaptic EPSCs with short and/or long latencies were evoked in SG neurons by stimulation of the dorsal root attached to spinal cord slices. These 2 types of monosynaptic responses were considered to be Aδ- or C-afferent-mediated responses based on the conduction velocity of the afferent fibers and stimulus threshold (Fig. 1B, C). The average conduction velocities of the afferent fibers were 6.5±0.4 m/sec for Aδ fibers (n=15) and 0.58±0.02 m/sec for C fibers (n=20). The threshold stimulus intensities for the monosynaptic Aδ fiber- and C fiber-evoked EPSCs were 0.10±0.02 mA (n=15) and 0.91±0.12 mA (n=20), respectively. These conduction velocities and stimulus intensities are comparable to those observed in previous studies [18,20,22].
The effects of 100 μM naftopidil on monosynaptic Aδ fiberand/or C fiber-evoked EPSCs were investigated using 26 SG neurons in total (Fig. 2). Each of the evoked EPSCs was reversibly inhibited in amplitude by naftopidil. When examined in many neurons, the inhibition of the peak amplitudes of Aδ fiber- and C fiber-evoked EPSCs was 72.0%±7.1% (P<0.001, n=15; Fig. 2A, B) and 70.0%±5.5% (P<0.001, n=20; Fig. 2A, C), respectively. DMSO in which naftopidil is dissolved is toxic to cells [23]. To exclude the possibility that the observed effect was mediated by DMSO, we examined the effects of DMSO alone on these neurons. DMSO at 1% showed no effect when present in the perfusate of Krebs solution on any of the Aδ fiber- and C fiber-evoked EPSCs tested compared with the control (Aδ fiber-evoked EPSCs; 98.1%±5.1% of control, P>0.05, n=2, C fiber-evoked EPSCs; 97.5%±1.7% of control, P>0.05, n=4). These results are consistent with our previous studies [16]. Although enhancement of spontaneous inhibitory transmission, which is produced by naftopidil in SG neurons, is not subject to desensitization [16], we also examined how repeated application of 100 μM naftopidil affected Aδ fiber- and C fiber-evoked EPSCs. As shown in Fig. 3A, additional naftopidil was administered at an interval of 20 minutes. The first and second naftopidil treatments produced similar decreases in Aδ fiberand C fiber-evoked EPSCs (Fig. 3). Relative to the control, Aδ fiber- and C fiber-evoked EPSC amplitudes in the presence of the initial application of naftopidil were almost comparable to the amplitudes after the second application (Fig. 3B), indicating that naftopidil activity was consistent with recovery from desensitization. Because 100 μM naftopidil repeatedly suppressed Aδ fiber- and C fiber-evoked EPSC amplitudes, we next investigated how prazosin, another α1-adrenoreceptor antagonist, affected this suppression in the same neurons. Bath application of 10 μM prazosin had no effect on any of the Aδ fiber- and C fiber-evoked EPSCs compared with the control (Aδ fiber-evoked EPSCs: 97.2%±1.8% of control, P>0.05, n=3; C fiber-evoked EPSCs: 97.2%±5.1% of control, P>0.05, n=4) (Fig. 4).
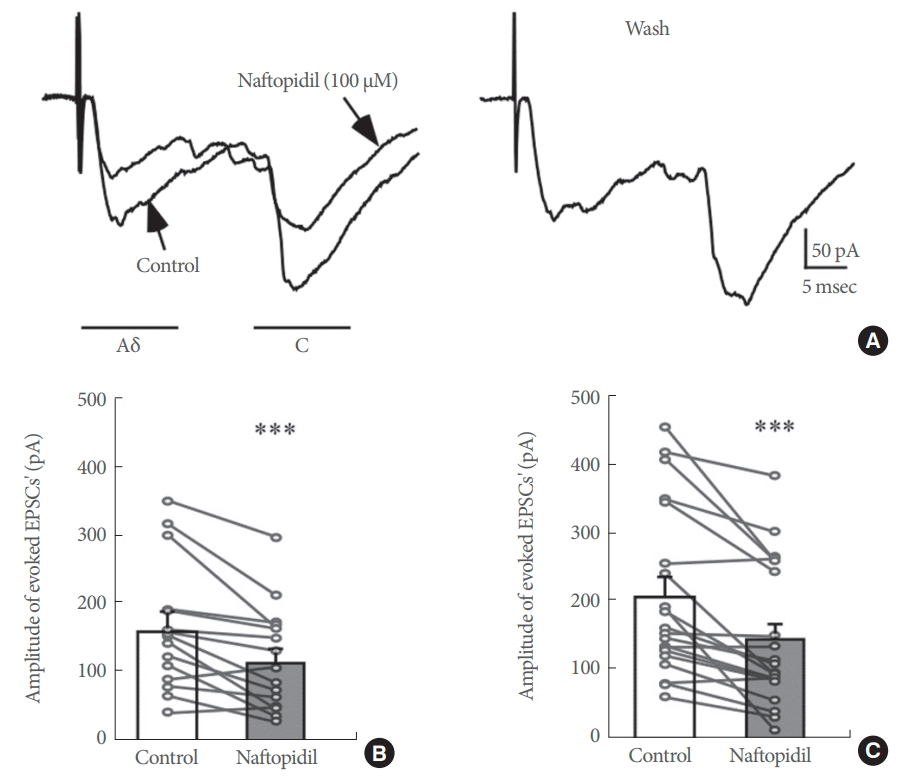
Effects of naftopidil on monosynaptic Aδ and C fiber-evoked excitatory postsynaptic currents (EPSCs) in substantia gelatinosa neurons. (A) Representative effects of 100 μM naftopidil on Aδ and C fiber-evoked EPSCs. (B) Summary of the effects of 100 μM naftopidil on the amplitude of Aδ fiber-evoked EPSCs. (C) Summary of the effects of 100 μM naftopidil on the amplitude of C fiber-evoked EPSCs. ***P<0.001 vs. control by paired t-test.
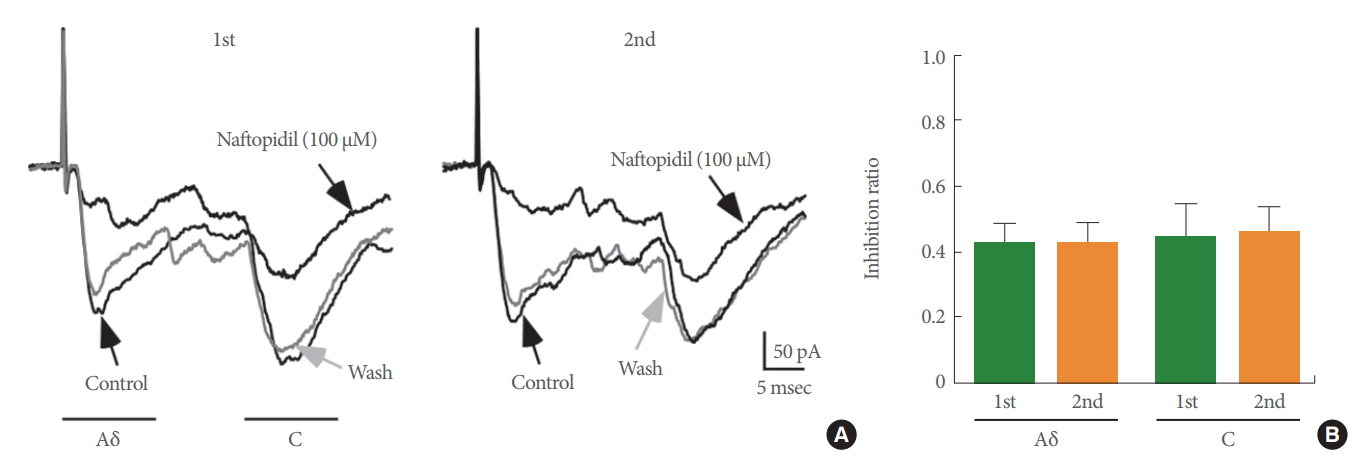
Naftopidil suppressed Aδ and C fiber-evoked excitatory postsynaptic current (EPSC) amplitudes in a repeated manner in a substantia gelatinosa neuron. (A) Chart recordings showing Aδ and C fiber-evoked EPSCs after the first and second applications of 100 μM naftopidil. (B) Summary of the effect of 100μM naftopidil on the amplitude of Aδ and C fiber-evoked EPSCs.

Naftopidil decreased the amplitude of Aδ and C fiber-evoked excitatory postsynaptic currents (EPSCs), but another α1-adrenoceptor antagonist, prazosin, did not. (A) Representative effects of 100 μM naftopidil and 10 μM prazosin on Aδ fiber-evoked EPSCs. (B) Representative effects of 100 μM naftopidil and 10 μM prazosin on C fiber-evoked EPSCs.
DISCUSSION
To evaluate the effects of naftopidil, an α1-adrenoceptor blocker, on evoked EPSCs, patch-clamp recording was performed in SG neurons from the spinal cord of adult rats. Naftopidil inhibited the amplitude of both monosynaptic Aδ fiber- and C fiber-evoked EPSCs, and these effects were reversible and reproducible. Using the same procedure, prazosin inhibited neither Aδ fiber- nor C fiber-evoked EPSCs at all.
Mechanism of Action of Naftopidil
In this study, the reduction in evoked EPSCs in the spinal cord from L5 to S1 by naftopidil indicates suppression of nociceptive stimulation or the micturition reflex. In a previous study, naftopidil increased the frequency of mIPSCs, but not their amplitude [16]. The frequency of mIPSCs in the presence of naftopidil was attenuated with bicuculline or strychnine. These data indicate that naftopidil may act on the synaptic terminals of inhibitory interneurons and enhance the release of glycine and GABA. Therefore, naftopidil may activate inhibitory interneuron terminals in the spinal dorsal horn. The amplitudes of both GABAergic-evoked and glycinergic-evoked IPSCs were also enhanced by naftopidil [16]. This result suggests that naftopidil may change the sensitivity of GABA and/or glycine receptors in the spinal dorsal horn neurons. However, the amplitude of mIPSCs was not affected following bath application of naftopidil. Consequently, these results exclude the possibility that naftopidil changes the sensitivity of GABA and/or glycine receptors in the spinal dorsal horn neurons. In addition, naftopidil inhibits the micturition reflex, and intrathecal bicuculline and/or strychnine reverses the inhibition [17]. Therefore, naftopidil seems to elevate the levels of GABA and/or glycine at the presynaptic site of SG neurons in the spinal cord. Consequently, evoked EPSCs may be inhibited by a reduction in glutamate release together with upregulation of GABA and/or glycine levels by naftopidil in SG neurons. This could contribute, at least in part, to the inhibitory modulation of somatosensory sensation in the SG by intrathecally administered naftopidil.
Alternatively, naftopidil may inactivate the influx of cations into SG neurons, subsequently suppressing the release of glutamate. This possibility is supported by our previous studies, as follows. We examined the actions of naftopidil on miniature EPSCs in adult SG neurons and showed that naftopidil increased the frequency of miniature EPSCs [16]. In the present study, naftopidil blocked monosynaptic Aδ fiber- and C fiber-evoked EPSCs. Similar to the case of transient receptor potential channels in a previous report [20,22,24], we propose that naftopidil depolarizes the presynaptic terminals or axons of Aδ and/or C afferents by activating naftopidil binding sites (not α1D/1A-adrenoceptors) expressed on the presynaptic membranes. This depolarizing action has a facilitatory effect on miniature EPSCs and may also inhibit evoked release from Aδ and C fibers through inactivation of voltage-gated Na+ channels or by shunting of Na+ currents [25].
Regarding the negative effect of prazosin (10μM), in addition to the findings of the previous studies mentioned above, naftopidil binds to human 5-HT2A and 5-HT2B receptors with pKi values of 6.55 and 7.82, respectively, and may functionally block the 5-HT2A and 5-HT2B receptors in the human urinary bladder [26]. However, outward current is induced in excitatory interneurons when 5-HT activates postsynaptic 5-HT1A receptors [27]. In contrast, in the small population of SG neurons, 5-HT induces an inward current mediated by the activation of postsynaptic 5-HT3 receptors on inhibitory interneurons [27,28]. Moreover, 5-HT inhibits glutamate release from C afferent fibers by activating presynaptic 5-HT1A-like receptors [29]. However, 5-HT1A, 2A, 2B, 2C, 3 receptor antagonists do not affect the excitatory and inhibitory synaptic response [27,29,30]. These results suggest that naftopidil must have acted on a binding site that does not bind prazosin. This binding site remains to be identified. However, previous research found that GABA and/or glycine receptors mediated the facilitation of mIPSCs and evoked IPSCs by naftopidil [16]. Therefore, GABAergic or glycinergic neurons may inhibit excitatory glutamatergic neurons. Finally, naftopidil may decrease excitation of the micturition reflex through such neural networks (Fig. 5).
Reversibility and Reproducibility
The effects of naftopidil on evoked EPSCs were reversible, suggesting that naftopidil may act on a binding site, likely its receptor. However, because prazosin did not inhibit evoked EPSCs, the effect of naftopidil on evoked EPSCs may not involve α1-adrenoceptors. In addition, naftopidil reduced evoked EPSCs in a reproducible manner, and therefore, naftopidil is not considered to produce desensitization of SG neurons, even at a high concentration.
Concentration of Naftopidil
Concentrations of naftopidil from 0.3 to 3 μM, 0.4 to 40 μM, and 0.5 to 100 μM have been used to determine detrusor contractility [26], platelet aggregation responses [31], and the viability of cancer cells [32,33], respectively. In the present study, a high concentration of 100 μM was used, although this concentration has also been used in previous studies. This concentration of 100 μM seems to be above that used clinically. However, the present results are probably useful for future studies, such as pharmacological studies of structure-activity correlations, the development of new drug delivery systems for local injection, and the development of derivatives with higher activity.
In conclusions, the present study suggests that naftopidil reduces the amplitude of evoked EPSCs, although its mechanism of action apparently does not involve α1-adrenoceptors. In addition to the facilitation of mIPSCs, as reported in a previous study, the inhibition of evoked EPSCs by naftopidil may also contribute to suppression of the micturition reflex together with nociceptive stimulation. Because these responses seem to be associated with analgesia, naftopidil or more highly active derivatives may be useful for treating LUTS with concomitant pain, such as interstitial cystitis/bladder pain syndrome [34].
Notes
Grant/Fund Support
This work was supported by grants from the programs for Grants-in-Aid for Scientific Research (DU and MY) of the Ministry of Education, Science, Sports and Culture of Japan (Grant Numbers JP15K08667, JP25860431, JP23790652, JP25460733, JP21600005), and was partially supported by the MEXT-Supported Program for the Strategic Research Foundation at Private Universities, “Creation of 3D nano-micro structures and its application to biomimetics and medicine,” 2015-2019 (Grant Number S1511031), and by The Hokugin Young Researcher Foundation (DU). This study was also supported in part by the Asahi Kasei Pharma Corporation.
Research Ethics
All experiments were carried out in accordance with the Guiding Principles for Care and Use of Animals in the Field of Physiological Sciences of the Physiological Society of Japan and were approved by the local Animal Experiment Committee of Kumamoto Health Science University and Kyushu University.
Conflict of Interest
Tsuyoshi Hattori is an employee of Asahi Kasei Pharma Inc. Except for that, no potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTION STATEMENT
·Full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis: DU
·Study concept and design: DU, MY
·Acquisition of data: DU
·Analysis and interpretation of data: DU, TH
·Drafting of the manuscript: DU, TH
·Critical revision of the manuscript for important intellectual content: MY
·Statistical analysis: DU, TH
·Obtained funding: MY
·Administrative, technical, or material support: TH
·Study supervision: MY

