Influence of Circadian Disruption Associated With Artificial Light at Night on Micturition Patterns in Shift Workers
Article information
Abstract
Shift workers often experience problems associated with circadian disruption associated with artificial light at night and nocturia is commonly noted in night-shift workers. Nocturia associated with circadian disruption is due to increased urine production of the kidney and decreased storage function of the bladder. A recent discovery of peripheral clock genes in the bladder and their role in contractile property of the bladder support that micturition is closely related to the circadian rhythm. Moreover, there are clinical studies showed that shift workers more often experienced nocturia due to circadian disruption. However, comparing with other health problems, concerns on nocturia and voiding dysfunction associated with circadian disruption are insufficient. Therefore, further studies about voiding dysfunction associated with the circadian disruption in shift workers are necessary.
INTRODUCTION
Artificial light at night (ALAN) changes and contributes to improve human’s lifestyle in modern world. Comparing with the past, nowadays, the amount of ALAN is tremendously increased as use of the various new devices such as computers, smartphones, and tablets has been common in daily life. Therefore, people in the present world are exposed to excessive light at night [1-3]. Exposure to ALAN has a harmful influence in human’s health and this undesirable effect is associated with the circadian disruption [4].
Several studies reported that ALAN exposure can decrease normal function of cognitive, metabolic, immune, and reproductive systems due to circadian disruption [5-7]. Well-known health issues associated with ALAN is obesity and metabolic diseases. A cross sectional study in United Kingdom showed positive correlation between obesity and light exposure at night [8]. Moreover, in vivo study with mice exposed to light at night demonstrated more weight gain and impaired glucose tolerance [5]. Cognitive impairment and mood disorder can be induced by ALAN due to excessive sleepiness at daytime and mood changes associated with sleep deprivation [9-11]. In addition, ALAN can increase risk of cancers such as breast, prostate, and colorectal cancers [12-15]. These adverse health effects are related with inhibition of night time secretion of melatonin induced by circadian disruption [16].
Common situation to expose to ALAN and disrupt circadian rhythm is shift work and about 15%–25% of people worked at night in modern world [17-19]. Shift workers experience more sleep deprivation, sleepiness, and insomnia and these sleep problems decrease working performance and increase errors and accidents. Moreover, health problems such as cognitive impairment, depression, metabolic diseases, cardiovascular diseases, and cancers comparing with nonshift workers [20]. Therefore, it is important to prevent and treat health problems associated with ALAN for shift workers.
Micturition is influenced by circadian rhythm and voiding dysfunction can be induced by circadian disruption. For example, nocturia is voiding dysfunction that people wake up to urinate during sleep and nocturia is induced by impairment of circadian antidiuretic hormone release and abnormal urine production at night. Decreased bladder capacity noted in people with abnormal sleep pattern is one of reason to induce nocturia compared with people with normal sleep pattern [21]. Therefore, people in shift work can experience voiding problems associated with circadian disruption and abnormal voiding is important risk factor to reduce well-being. However, concern about abnormal voiding is insufficient compared with other health issues associated with shift workers. This review explores voiding dysfunction found in shift workers based on investigational and clinical studies.
ROLE OF CLOCK GENE IN THE BLADDER
Circadian rhythm is controlled by master circadian clock, located in the suprachiasmatic nucleus (SCN) of the hypothalamus and circadian clock oscillations, at the molecular level, are governed by transcription-translation feedback loops. The feedback loop is consisted of core clock genes such as circadian transcription factor complex (CLOCK) and brain and muscle Arnt-like 1 (BMAL1) that positively interacts with the promoters of clock-controlled genes and proteins period (Per) and cryptochrome like protein (Cry) that inhibit their own transcription by interacting with the CLOCK/BMAL1 complex [22,23]. Moreover, molecular clock is existed in the various peripheral organs and coordination between central clock in SCN and peripheral clock in each organ maintains physiological function [24].
Normal voiding is influenced by urine production of the kidney and urine storage of the bladder and changes of production and storage of urine are regulated by circadian control. Increased plasma vasopressin modulated by central circadian clock at late sleep period reduces urine production by water reabsorption and electrolyte in the kidney. There are animal studies to show that how circadian disruption influence urine production of the kidney at molecular level. A study using Clock knockout mice showed that loss of circadian rhythm of renal sodium excretion and plasma aldosterone level [25]. Noh et al. [26] reported that mice deficient in Per1 and Per2 lost diurnal changes of urine volume compared with wild type mice. In addition, there are studies to show existence of clock genes in the bladder. Multiple genetic oscillators such as Per1, Bmal1, and Rev-erbα were found in the bladder and expression of clock genes were influenced by light-dark cycle. A recent study in CLOCK gene mutant mice showed that increased number of voiding and urine volume during sleep phase compared with wild type mice [27]. These findings meant that CLOCK gene deficiency in both of the bladder and kidney by decreased storage function of the bladder and increased urine production of the kidney at night.
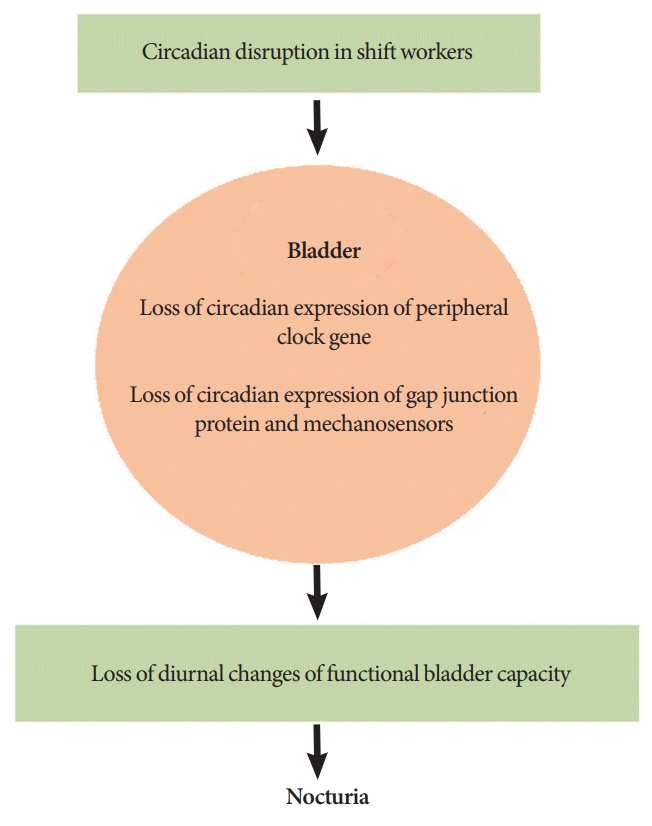
These findings proved the existence of peripheral clock in the bladder and, therefore; the next step is to identify the actual role of clock gene in the storage function of the bladder. Storage function of the bladder is regulated by contraction and relaxation of bladder smooth muscle and various factors are involved in increase or decrease of bladder capacity [28]. Moreover, diurnal changes of contractile properties of the bladder smooth muscle was noted [29]. Negoro et al. [30] found diurnal variation of connexin 43 (Cx43) of the bladder smooth muscle and involvement of peripheral clock in the bladder. They focused on Cx 43 because several studies showed that Cx43 was a gap junction protein existed in the bladder smooth muscle and was associated with bladder contraction. Gap junctions composed of Cx43 enables cell-to-cell signaling. Therefore, increased cell-to-cell signaling through gap junctions containing Cx43 induces intercellular electrical signaling and these changes can produce conditions such as overactive bladder (OAB) that decreased storage function of the bladder [31-33]. According to the results by Negoro et al. [30], diurnal variation of Cx43 in the bladder smooth muscle is coordinated with circadian clock of the bladder. Thus, diurnal variation of Cx43 associated with clock gene may contribute increased bladder capacity during sleep and it enhances bladder storage function to prevent nocturia at night.
Urothelium, mucosal layer of the bladder, also plays a role to control storage function of the bladder. Urothelium is the first tissue to sense bladder extension when the bladder is filled with urine and activation of afferent pathway transmits signals in response to stretch of the bladder wall. These afferent signals induce bladder contraction and reduce bladder capacity [34,35]. Based on this role of urothelium, there is a recent study to find the association of mechanosensor of urothelium and circadian clock. After bladder extension, mechanosensors of the urothelium induce releasing neurotransmitters, adenosine triphosphate (ATP). Mechanosensors in urothelium such as transient receptor potential cation channel subfamily V member 4 (TRPV4) and Piezol1 trigger ATP release in response to the bladder expansion [36-38]. Also, vesicular nucleotide transporter (VNUT) and gap junction composed of connexin 26 (Cx26) in urothelium plays a role in ATP release [33,39,40]. Ihara et al. [41] showed diurnal changes of urothelial mechanosensors (TRPV4 and Piezol1), VNUT, and Cx26 that increased during the active period and decreased during the sleep period. These changes were coordinated with the diurnal expression of clock gene of the urothelium. However, not only the disruption of clock gene expression but also loss of diurnal changes of mechanosensors, VNUT, and Cx26 were observed in mice deficient with clock gene. Therefore, mice deficient with clock gene showed increased sense of the bladder extension and their bladder capacity during sleep period was decreased compared with normal mice. And this hypersensitivity to bladder extension due to disruption of circadian rhythm may reduce storage function of the bladder and induce nocturia.
CHANGE OF VOIDING PATTERN IN SHIFT WORKERS
Although it is well known that urine production and bladder capacity are influenced by circadian rhythm, studies about the prevalence and characteristics of voiding problems in shift workers are insufficient. Previous studies revealed that nurses often experienced voiding dysfunction such as nocturia, OAB and urinary incontinence (UI) compared with general population. And they found that voiding dysfunction is an important factor to reduce quality of life (QoL) and productivity at working place [42,43]. However, the previous investigators did not focus on the influence of circadian disruption. Instead, they concluded that voiding dysfunction was resulted from occupational stress and inconvenient environment to induce poor bladder habits. A recent study also showed that severe UI was associated with intentions to stop in nurses and midwifes, however; relationship between circadian disruption and severe UI could not analyzed because the study included workers for part-time or days only (not shifts) [44]. In general, most of the studies about the prevalence, impact, and cause of voiding dysfunction in working population seem to be lack of quality because they usually included only women and there were no occupational diversities [45].
There were 2 recent prospective studies to investigate voiding patterns in shift workers. Kim et al. [46] compared voiding patterns between nurses worked as rotational shifts and regular daytime. Comparing with nurses with regular daytime, nurses worked as rotational shifts showed increased urine volume per day (both day and night) and mean voiding volume was significantly increased during night shift. Moreover, nocturia was increased during night shift. After return to day shift, urine volume was decreased, however; urine volume at night was not decreased and nocturia was not improved. These findings showed that circadian disruption due to shift work induced urine production at night and nocturia. However, the limitation of the report by Kim et al. [46] was small number of participants (nurses worked as rotational shift: n=19 and nurses worked as regular daytime: n=7). Later, the other study including large number of shift workers (n=365; 200 males, 165 females) analyzed the effect of circadian disruption on nocturia [47]. The number of nocturia was higher in night-shift workers compared with nonshift workers. Urine volume at night of night-shift workers was not significantly different from nonshift workers. However, significantly reduced functional bladder capacity was observed in night-shift workers. Thus, the author suggested that nocturia of night-shift workers was induced by decreased storage function of the bladder rather than increased urine production at night. Although functional bladder capacity between nurses worked as rotational shift and regular daytime was not significantly different from the study by Kim et al. [46], similar change of the mean voided volume reflecting decreased storage function of the bladder at night was observed in rotational-shift nurses. However, in case we focused functional bladder capacity during night time, nurses worked as rotational shift showed significantly decreased functional bladder capacity during night shift compared with day shift. Therefore, circadian disruption in shift workers induces nocturia by increased urine production of the kidney and decreased storage function of the bladder (Fig. 1).
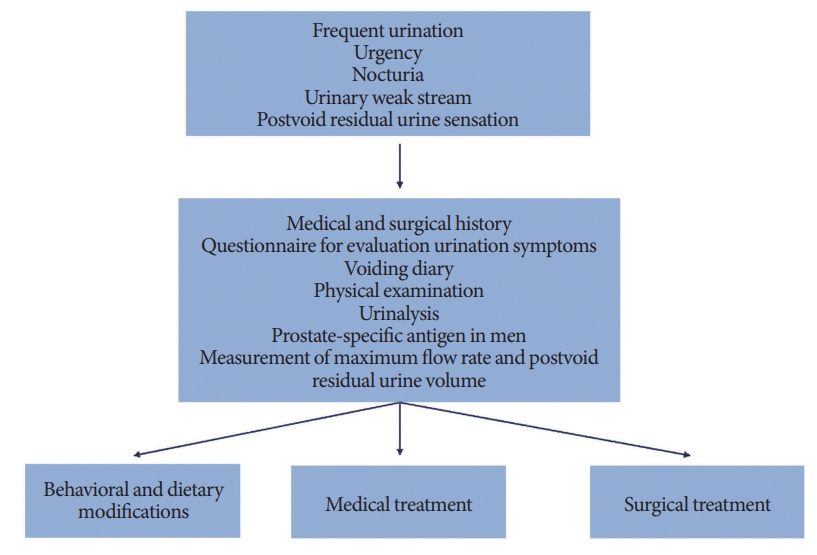
Underlying mechanism to induce voiding dysfunction observed in the shift workers is different from others, however, shift workers with voiding problem showed common symptoms such as frequent urination, urge to void, and nocturia. Therefore, general approach for the diagnosis and treatment of voiding dysfunction can be applied for the shift workers (Fig. 2) [48-53].
SUMMARY AND CONCLUSIONS
Micturition is a human behavior closely associated with circadian rhythm and several recent studies showed the existence of peripheral clock in the bladder and their role in the control of bladder function. However, well-designed clinical study to present the prevalence, impact, and characteristics observed in shift workers are lacking comparing the importance of voiding dysfunction associated with circadian disruption. Moreover, at present, there are studies concerning only nocturia of shift workers and attention about voiding patterns of daytime has been relatively insufficient. Therefore, further well-designed clinical studies are necessary to understand circadian disruption associated voiding problems and improve QoL of shift workers experiencing voiding dysfunction.
Notes
Fund/Grant Support
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2018R1D1A3B07048492). This work was supported (in part) by the Yonsei University Wonju Campus Future-Leading Research Initiative of 2018 (2018-62-0039). This study was supported by the Ministry of Education of the Republic of Korea and the National Research Foundation of Korea (NRF-2017R1A2B2005412). Basic Science Research Program through the National Research Foundation of Korea which was funded by the Ministry of Education supported and funded this work (NRF-2015R 1D 1A 1A 01060982).
Conflict of Interest
SJK, JWK, YSC, and KHK, members of the Editorial Board of International Neurourology Journal, are the authors of this article. However, they played no role whatsoever in the editorial evaluation of this article or the decision to publish it. Except for that, no potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTION STATEMENT
·Full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis: KHK, SJK
·Study concept and design: SJK, KHK
·Acquisition of data: JWK, YSC
·Analysis and interpretation of data: SJK, JWK
·Drafting of the manuscript: SJK
·Critical revision of the manuscript for important intellectual content: HNY
·Obtained funding: SJK, KJC, KHK
·Administrative, technical, or material support: JWK, YSC
·Study supervision: KHK